Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter7: Components Of Optical Instruments
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Q. based on the graph, the absorbance of caffeine using UV-vis spectroscopy is at ________.
CHOICES:
| A. |
320 nm |
| B. |
260 nm |
| C. |
260-290 nm |
| D. |
275 nm |
| E. |
260-300 nm |
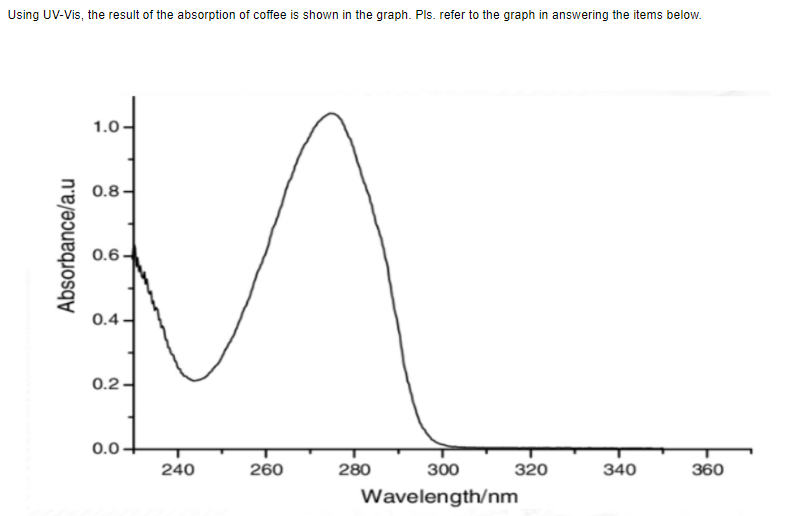
Transcribed Image Text:Using UV-Vis, the result of the absorption of coffee is shown in the graph. Pls. refer to the graph in answering the items below.
1.0-
0.8
0.6
0.4
0.2-
0.0
240
260
280
300
320
340
360
Wavelength/nm
Absorbance/a.u
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning
