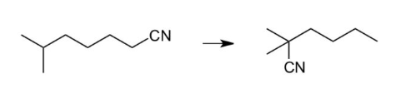
Some bond dissociation energies in kJ/mol are listed. 1°, 2°, 3° means primary, secondary, tertiary. 1° C-H 423 2° C-H 413 3° C-H 404 1° C-CN 509 2° C-CN 506 3° C-CN 493 For the hypothetical reaction below, do two things: 1) Estimate the enthalpy change ΔH° for the reaction. Show your work in the space below. 2) Draw a circle around the molecule with the lower enthalp
Bond Parameters
Many factors decide the covalent bonding between atoms. Some of the bond parameters are bond angle, bond order, enthalpy, bond length, etc. These parameters decide what kind of bond will form in atoms. Hence it is crucial to understand these parameters in detail and understand how changing these parameters affects the kind of bonding or various characteristics.
Bond Dissociation Energy
The tendency of an atom to attract an electron is known as its electronegativity.
Some
tertiary.
1° C-H 423 2° C-H 413 3° C-H 404
1° C-CN 509 2° C-CN 506 3° C-CN 493
For the hypothetical reaction below, do two things:
1) Estimate the enthalpy change ΔH° for the reaction. Show your work in the space below.
2) Draw a circle around the molecule with the lower enthalpy.
Step by step
Solved in 3 steps with 1 images









