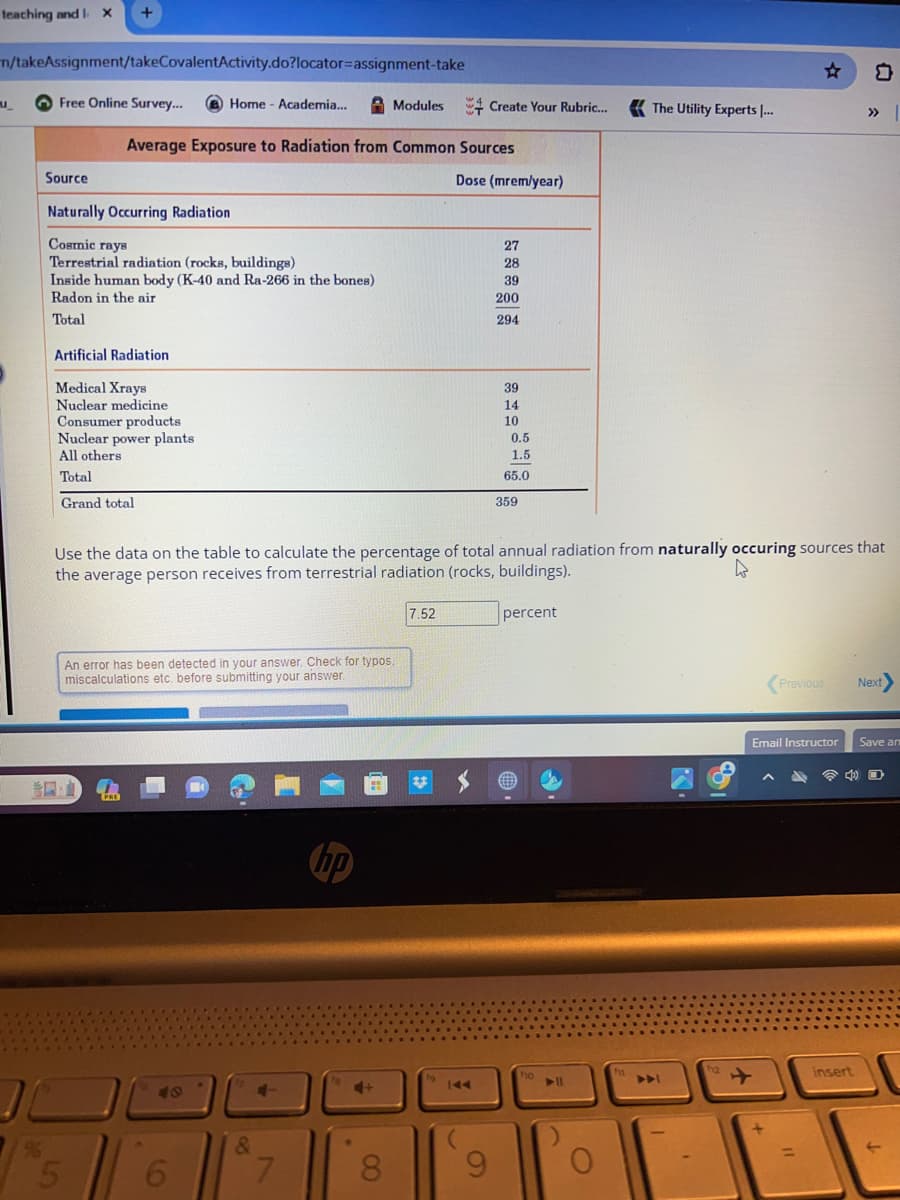
Source Average Exposure to Radiation from Common Sources Naturally Occurring Radiation Cosmic rays Terrestrial radiation (rocks, buildings) Inside human body (K-40 and Ra-266 in the bones) Radon in the air Total Artificial Radiation Medical Xrays Nuclear medicine Consumer products Nuclear power plants All others Total Grand total Dose (mrem/year) 7.52 27 28 39 200 294 39 14 10 0.5 1.5 65.0 359 Use the data on the table to calculate the percentage of total annual radiation from naturally occuring sources that the average person receives from terrestrial radiation (rocks, buildings). percent
Source Average Exposure to Radiation from Common Sources Naturally Occurring Radiation Cosmic rays Terrestrial radiation (rocks, buildings) Inside human body (K-40 and Ra-266 in the bones) Radon in the air Total Artificial Radiation Medical Xrays Nuclear medicine Consumer products Nuclear power plants All others Total Grand total Dose (mrem/year) 7.52 27 28 39 200 294 39 14 10 0.5 1.5 65.0 359 Use the data on the table to calculate the percentage of total annual radiation from naturally occuring sources that the average person receives from terrestrial radiation (rocks, buildings). percent
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter9: Nuclear Chemistry
Section: Chapter Questions
Problem 9.50P
Related questions
Question
Transcribed Image Text:teaching and 1. x
n/takeAssignment/takeCovalentActivity.do?locator=assignment-take
u_
Free Online Survey...
+
Source
Naturally Occurring Radiation
Cosmic rays
Terrestrial radiation (rocks, buildings)
Inside human body (K-40 and Ra-266 in the bones)
Radon in the air
Total
Artificial Radiation
Medical Xrays
Nuclear medicine
Consumer products
Nuclear power plants
All others
Total
Grand total
70
9
Average Exposure to Radiation from Common Sources
5
PRE
Home - Academia...
An error has been detected in your answer. Check for typos.
miscalculations etc. before submitting your answer.
48
6
***
&
Use the data on the table to calculate the percentage of total annual radiation from naturally occuring sources that
the average person receives from terrestrial radiation (rocks, buildings).
4
4-
7
hp
Modules Create Your Rubric...
•
4+
8
Dose (mrem/year)
7.52
27
28
39
200
294
9
39
14
10
0.5
1.5.
65.0
359
percent
ho
O
The Utility Experts ...
f11
▶▶
A
f12
Previous
Email Instructor
❤
insert
Next>
Save an
O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning