Spectrum No.3 The infrared spectrum of this compound shows abroad peak at 3370 cm³¹. There is also a strong peak at 1159 cm³¹, the mass spectrum shows no molecular ion peak. You will have to deduce the molecular weight from the heaviest fragment peak which arises from the loss of a methyl group from the molecular ion. Give the mechanism that lead to the formation of ion fragment at m/z= 59
Spectrum No.3 The infrared spectrum of this compound shows abroad peak at 3370 cm³¹. There is also a strong peak at 1159 cm³¹, the mass spectrum shows no molecular ion peak. You will have to deduce the molecular weight from the heaviest fragment peak which arises from the loss of a methyl group from the molecular ion. Give the mechanism that lead to the formation of ion fragment at m/z= 59
Chapter12: Structure Determination: Mass Spectrometry And Infrared Spectroscopy
Section12.SE: Something Extra
Problem 49AP: The infrared spectrum of the compound with the mass spectrum shown below has a medium-intensity peak...
Related questions
Question
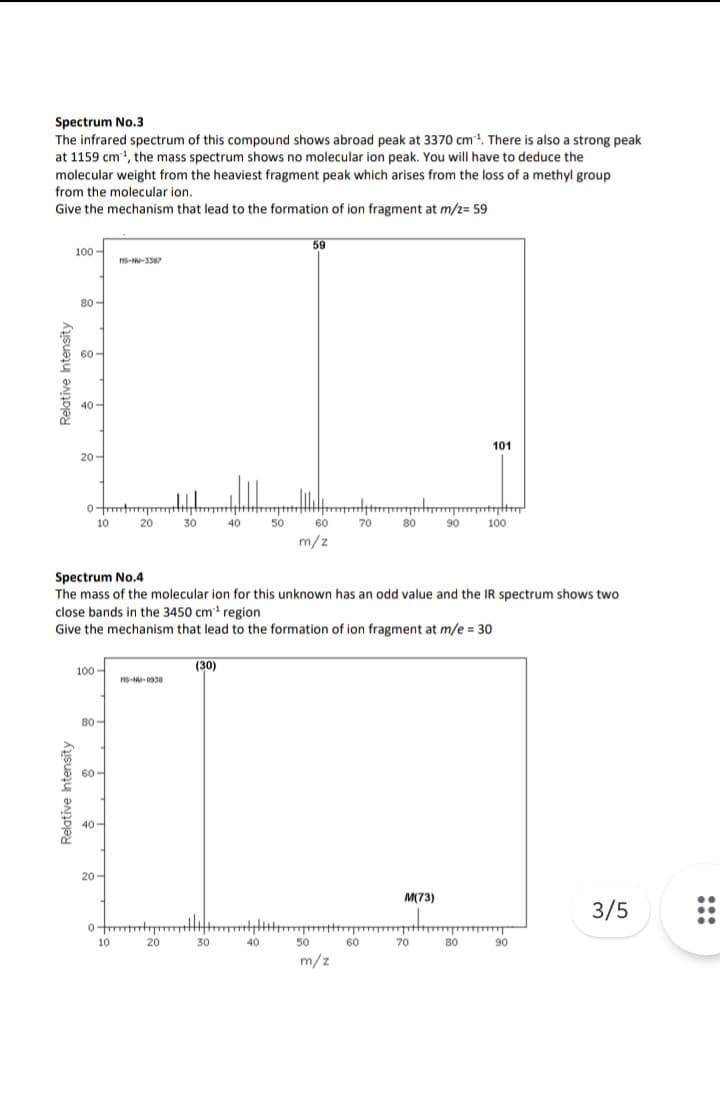
Transcribed Image Text:Spectrum No.3
The infrared spectrum of this compound shows abroad peak at 3370 cm ¹. There is also a strong peak
at 1159 cm¹, the mass spectrum shows no molecular ion peak. You will have to deduce the
molecular weight from the heaviest fragment peak which arises from the loss of a methyl group
from the molecular ion.
Give the mechanism that lead to the formation of ion fragment at m/z= 59
Relative Intensity
100-
Relative Intensity
80-
60-
40-
20
0-mmmmm
10
100-
80
60-
40
18-N-3387
20-
20
15--0930
0-T
10
30
20
Spectrum No.4
The mass of the molecular ion for this unknown has an odd value and the IR spectrum shows two
close bands in the 3450 cm³¹ region
Give the mechanism that lead to the formation of ion fragment at m/e= 30
(30)
40
30
50
40
59
60
m/z
50
m/z
70
60
80
M(73)
90
70
101
80
rytter
100
90
3/5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning