Spin-spin coupling between two hydrogen nuclei is mediated through chemical bonds. In the ¹H NMR, this coupling is usually observed through neighboring C-H bonds…. Does this means 1 bond???
Spin-spin coupling between two hydrogen nuclei is mediated through chemical bonds. In the ¹H NMR, this coupling is usually observed through neighboring C-H bonds…. Does this means 1 bond???
Chapter95: Nuclear Magnetic Resonance Spectroscopy (proton Nmr)
Section: Chapter Questions
Problem 6P
Related questions
Question
Spin-spin coupling between two hydrogen nuclei is mediated through chemical bonds. In the ¹H NMR, this coupling is usually observed through neighboring C-H bonds…. Does this means 1 bond???
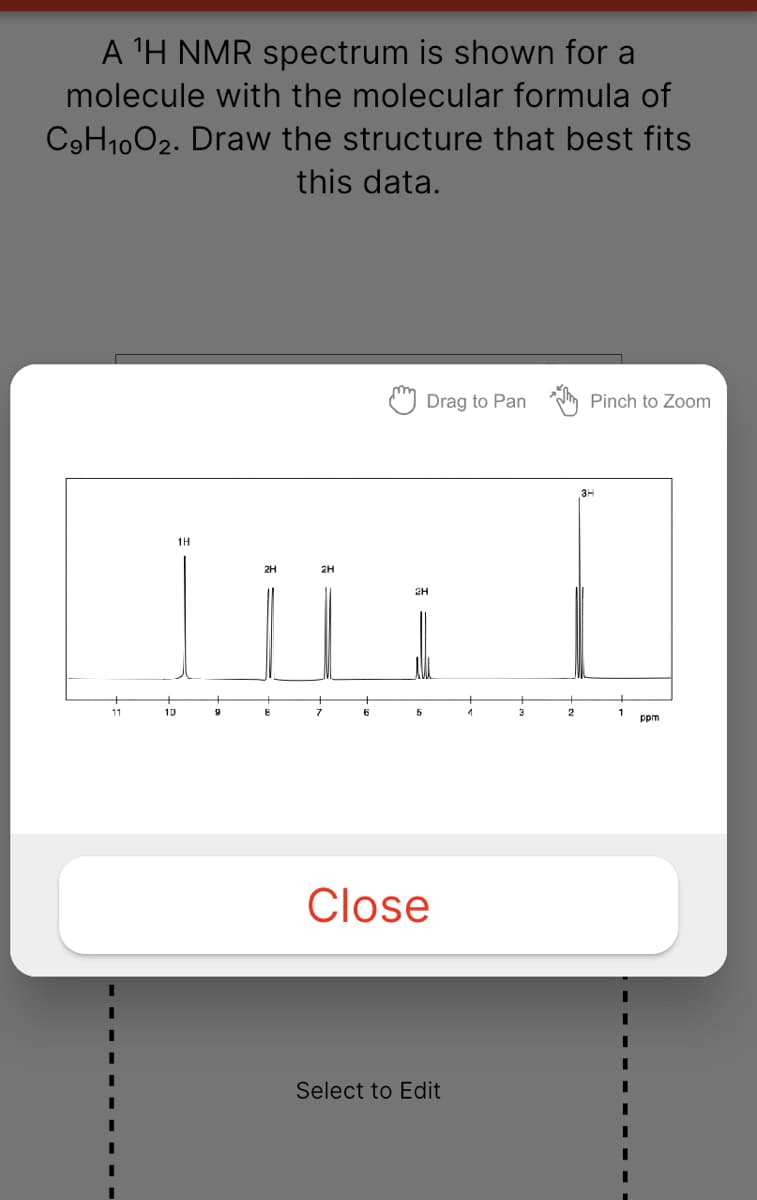
Transcribed Image Text:A 'H NMR spectrum is shown for a
molecule with the molecular formula of
C9H1002. Draw the structure that best fits
this data.
Drag to Pan
Pinch to Zoom
1H
2H
2H
2H
11
13
2
ppm
Close
Select to Edit
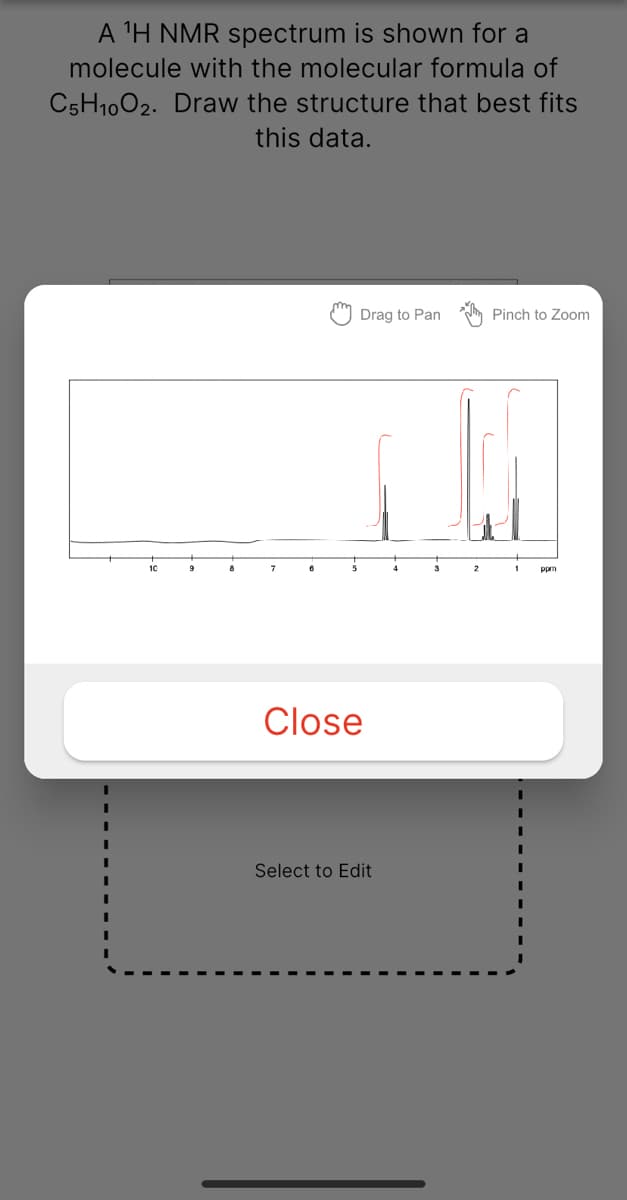
Transcribed Image Text:A 'H NMR spectrum is shown for a
molecule with the molecular formula of
C5H1002. Draw the structure that best fits
this data.
Drag to Pan
Pinch to Zoom
10
ppm
Close
Select to Edit
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT