statement regarding kinetic energy is true? sc.7.P.112 A The kinetic energy of an object is A O calculated by multiplying mass times speed. B The objects at rest will have more В Icinotia onormu than the
statement regarding kinetic energy is true? sc.7.P.112 A The kinetic energy of an object is A O calculated by multiplying mass times speed. B The objects at rest will have more В Icinotia onormu than the
Chapter1: Numerals And Fractions
Section: Chapter Questions
Problem 2PP
Related questions
Question
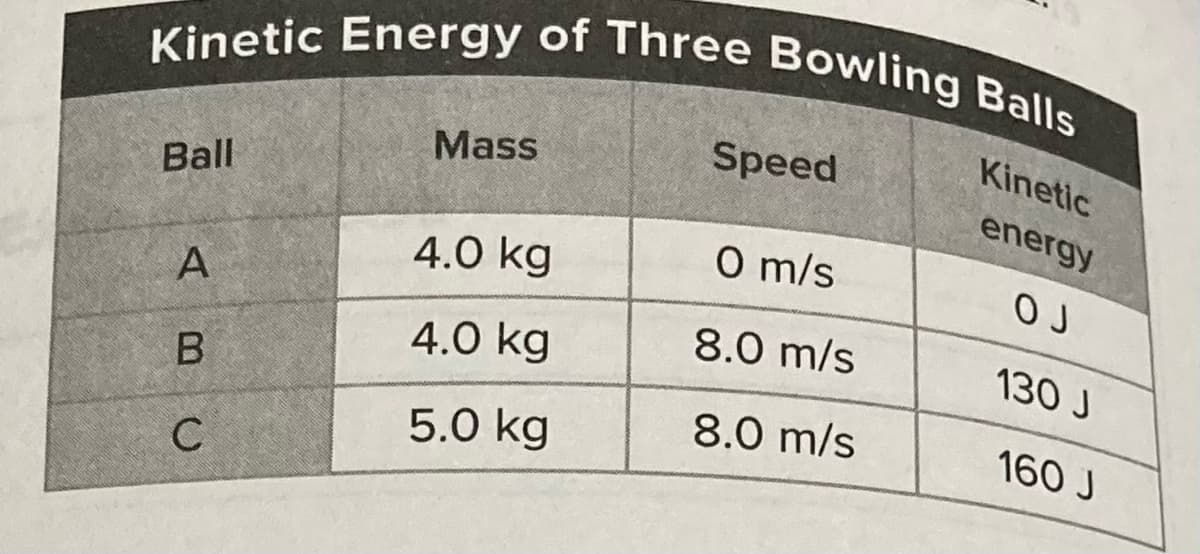
Transcribed Image Text:Kinetic Energy of Three Bowling Balls
Kinetic
Mass
Speed
Ball
energy
4.0 kg
O m/s
A
4.0 kg
8.0 m/s
130 J
B
5.0 kg
8.0 m/s
160 J
C

Transcribed Image Text:regarding kinetic energy is true? sc.7.P.11.2
Based on the table above, which statement
regarding kinetic energy is true? sc.7.P112
A The kinetic energy of an object is
A
O calculated by multiplying mass times
speed.
B The objects at rest will have more
kinetic energy than the objects in
В
motion.
© Objects that have the same mass have
the same kinetic energy no matter how
fast the object moves.
When objects travel at the same speed,
D
the more massive object has more
kinetic energy.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you







Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning