Q: Compound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be…
A:
Q: Consider the following reaction (DPPE structure below): F. - B(OH)2 F Ni(OTf)2 DPPE K2CO3 (a) Give…
A: The name reaction of the given transmetalation is known as Suzuki-Miyaura coupling in which metal…
Q: An optical core righyende N, when completely hydolysed yields two moles of myrsic acids (140) an one…
A: Triglycerides are tri esters formed when a glycerol molecule is bonded to three fatty acids with…
Q: An unknown hydrocarbon G, whose formula is C16H26 contains two triple bonds. Ozonolysis followed by…
A: Ozonolysis: Ozonolysis is an organic reaction where the unsaturated bonds of alkenes, alkynes,…
Q: To prepare butanoic acid from 1-propanol, which sequence of reagent(s) is/are best employed? A…
A: We will do series of reactions and then get required product.
Q: Explain and describe objective for preparation of methyl orange?
A: Methyl orange is a pH indicator. It has two distinct forms. Red in acidic medium and yellow in basic…
Q: Illustrate the cope elimination is an intramolecular E2 of tertiary amine oxides
A: The intramolecular elimination of tertiary amine oxide to form corresponding alkenes on heating is…
Q: A mono-acid organic base gave the following results on analysis. C 78.6% H = 84% N = 13%. 0.4 gm of…
A:
Q: о || || НСНО, PҺCHO, С,Н, - С-сH,, СН, - С-Н, Н-С-С-н о || || Ph-C-С-Н
A: Aldehydes and ketones are carbonyl compounds that undergo characteristic, nucleophilic addition…
Q: . Give the product for the tranformation given below. (Hint selective oxidation)
A: Oxidation of alcohols : In general 1o alcohols upon oxidation gives aldehyde with mild…
Q: When pyrrole is added to a dilute solution of D2SO4 in D2O, 2-deuteriopyrrole is formed. Propose a…
A: The mechanism for the formation of 2-deuteriopyrrole has to be given,
Q: Which of the following statements is TRUE about elimination tests for anions? Select one: O a. NO;…
A: Anionic analysis refers to identification of anions present in the salt mixture. Various anions…
Q: carbon and 345% hydrogen on the molar fonmula of the compound? (Relative atomic mass C-12.0, O-16.0,…
A: Given:- Molar mass of compound = 116 Relative composition of Carbon = 41.38% Relative composition…
Q: 4.Give two 4spets ook at the' structure of ad that need to be fultilled when made dye by diazo you…
A: These are generally made from primary aromatic amine in addition to nitrous acid.
Q: spectru moleculor formula and use the formula to calculate units of ursaturation Assume that the…
A: From this we will do the calculation of the molecular formula of the compound who gives this…
Q: Adipic acid is an important bifunctional molecule that can be Но, used as a linker between proteins…
A:
Q: How would How would you Seperate A, B, and C below and they were mixed using eanipment. an…
A: The answer is as follows:
Q: Writing a presentation entitled Hydrogenation with some for its introduction and then explaining
A: Following are the introduction and explanation for hydrogenation.
Q: suggest a synthesis of this product from Geranylgeranylpyrophosphate.. and what is the name of the…
A:
Q: Which of the following is/are TRUE in the experiment about SUDAN-1? 1. The preparation of…
A: Answer : I, II, IV True statements : I. The preparation of phenyldiazonium ion was carried out at…
Q: Outline the synthesis of racemic 3-methyl-3-heptanol, Et(Me)C(*)OHn-Bu, starting from alcohols of…
A: Using a 4 or less membered alcohol, we have to form 3-methyl 3-heptanol.
Q: Question 4
A: Nucleophiles are those species which act as electron rich. These species offer electrons during any…
Q: The triple amine in an acidic medium does not give a transition n→o * Explain why?
A:
Q: The nitration of the 2-amine toluene is able to occurs with only nitric acid. Interestingly it…
A:
Q: 5. Provide all the missing products (A-C) and reagent (i). Suggest retrosynthetic analysis for this.…
A:
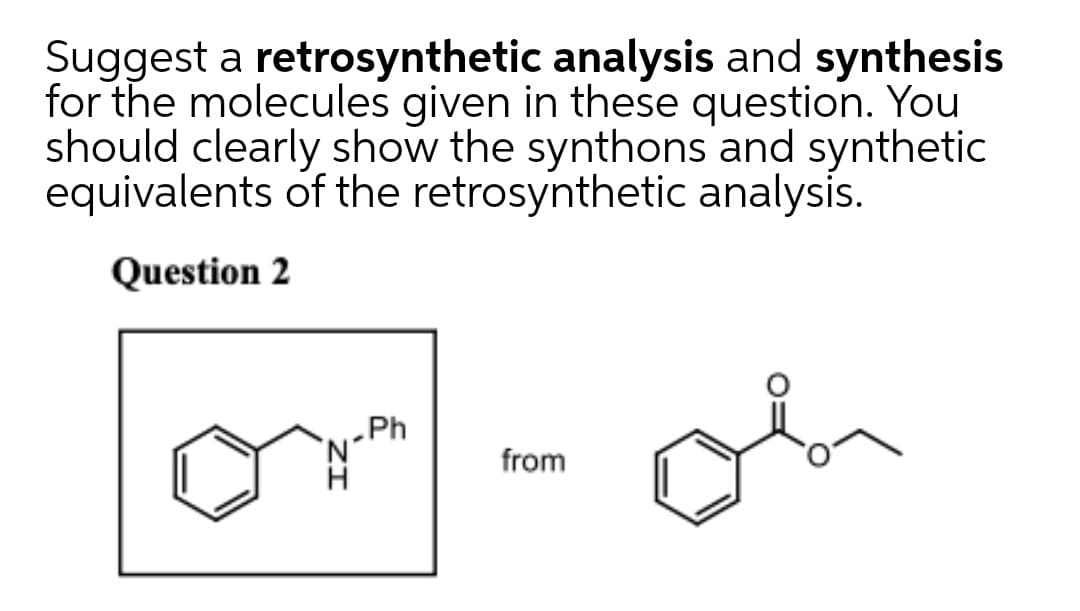
Q: Suggest Syntheses for the following from the designated starting materials and any other necessary…
A:
Q: Compound K, L and M are three isomers with the molecular formula C5H10O. Compound K cannot be…
A: All the details are explained in handwritten solution.
Q: In F. Dean Toste's 2007 total synthesis of (+)-Fawcettimine (Angew Chem, 46 (40), 7671-7673), a…
A: The correct missing components form the two boxes are as follows;'
Q: OMe OMe
A:
Q: A compound E C7H12O gives a crystalline precipitate with 2,4-dinitrophenylhydrazine. It gives a…
A:
Q: 17.4 Give explanations for the following observations. (a) pK, values for CF;CO,H and CH3CO,H are…
A: Given,
Q: What is procedure in extraction of beta-carotene from carrot.
A: We have to write the procedure for extraction of beta carotene from carrot.
Q: Suggest a method of preparing the following compound from tert-butoxybenzene (NB. use as many steps…
A: Electrophilic aromatic substitution is a reaction in which electrophile replaces a hydrogen atom of…
Q: to write ionic equation for the reaction of acetic acid with itheim bromide, and specify Rwhether…
A: Starting materials Acetic acid : CH3COOH Lithium bromide : LiBr pKa of acetic acid = 4.7 pKa of…
Q: 39), Thionyl chaloride, sOcz orying ageet in meay syotetic chamistry experimepts isi used as a very…
A:
Q: Tertiary aromatic amine reacts with NaNO2 / HCI at (0 C Oto give deep brown precipitate of: * a.…
A: Answer
Q: Identify the functional groups
A: In this question we have to identify the functional groups present in each drug and the drup…
Q: Predict the products of the following reactions AICI, A. AICI, B. C. Nitration of bromobenzene D.…
A: Complete the following reaction
Q: TRELAB EXE. E: Write balanced equations for the dichromate and hypochlorite oxidations of…
A:
Q: 3. Draw the structure of procainamide hydrochloride, mark the aromatic amino-group and suggest the…
A: Procainamide is an aromatic amide group. The molecule looks as follows:
Q: The reaction between Pl3, PSCl3 and Zn powder gives P3ls as one of the products. The sołution state…
A: This Question is based upon P31 NMR.
Q: Question 5
A: Knoevenagel condensation is a type of organic reaction in which an active hydrogen containing…
Q: A Student added 1.5 ml of 3 M Methyl Magnesium Bromide to a conical vial containing Benzophenone &…
A: Methyl magnesium bromide acts as a good source of Nucleophile CH3- , thus it carry out nucleophilic…
Q: In an essay provide written explana tion, In the synthesis of Dibenzalacetone, why must there be 2x…
A:
Q: Using the trans effect sequence given in the text, devise rational procedures for selectively…
A: Trans effect is defined as the ability of a group for directing the incoming group trans to it's…
Step by step
Solved in 2 steps with 1 images

- When cyclohexanol is dehydrated to cyclohexene, a gummy green substance forms onthe bottom of the flask. Suggest what this residue might be, and propose a mechanismfor its formation (as far as the dimer).Suggest ways of synthesizing the following compounds, how would youmake the disconnections and what are the synthons?Suggest ways of synthesizing the compound, how would youmake the disconnections and what are the synthons?
- Electrophilic nitration of benzoic acid gives almost exclusively 3-nitrobenzoic acid. By drawing the appropriate resonance forms of the intermediate cations resulting from attack of [NO2]+, explain this result.(19, 8, 3) Specify a synthetic scheme that would produce the compound shown above in the fewest steps possible. Use one of the starting materials shown together with any of the available reagents. Give the number of the starting material followed by the letters of the reagents in the order of their use, for example: 3be.Give the reagents and conditions to provide the following transformations, usethe 1. 2. etc. notations as appropriate
- 3. Benzalacetophenone can be nitrated to give the mononitro derivative.(a) Provide the structure of the mononitro derivative.(b) If a second nitro group is introduced, where would it enter? Give the structure of theproduct.Starting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to synthesize the following. Assume in all syntheses that mixtures of ortho-para products can be separated into the desired isomer. Q.)p-Chlorobenzoic acidGiven this retrosynthetic analysis, show how meclizine can be synthesized from the four named organic starting materials.
- Sketch and explain the IR, UV, mass, and NMR (1H and 13C) spectroscopy data of BenzonitrileWrite the synthesis of the target compounds by starting from each benzene given in the following items and by writing the mechanism of each step separetely.Starting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to synthesize the following. Assume in all syntheses that mixtures of ortho-para products can be separated into the desired isomer. Q.)m-Nitrobenzenesulfonic acid

