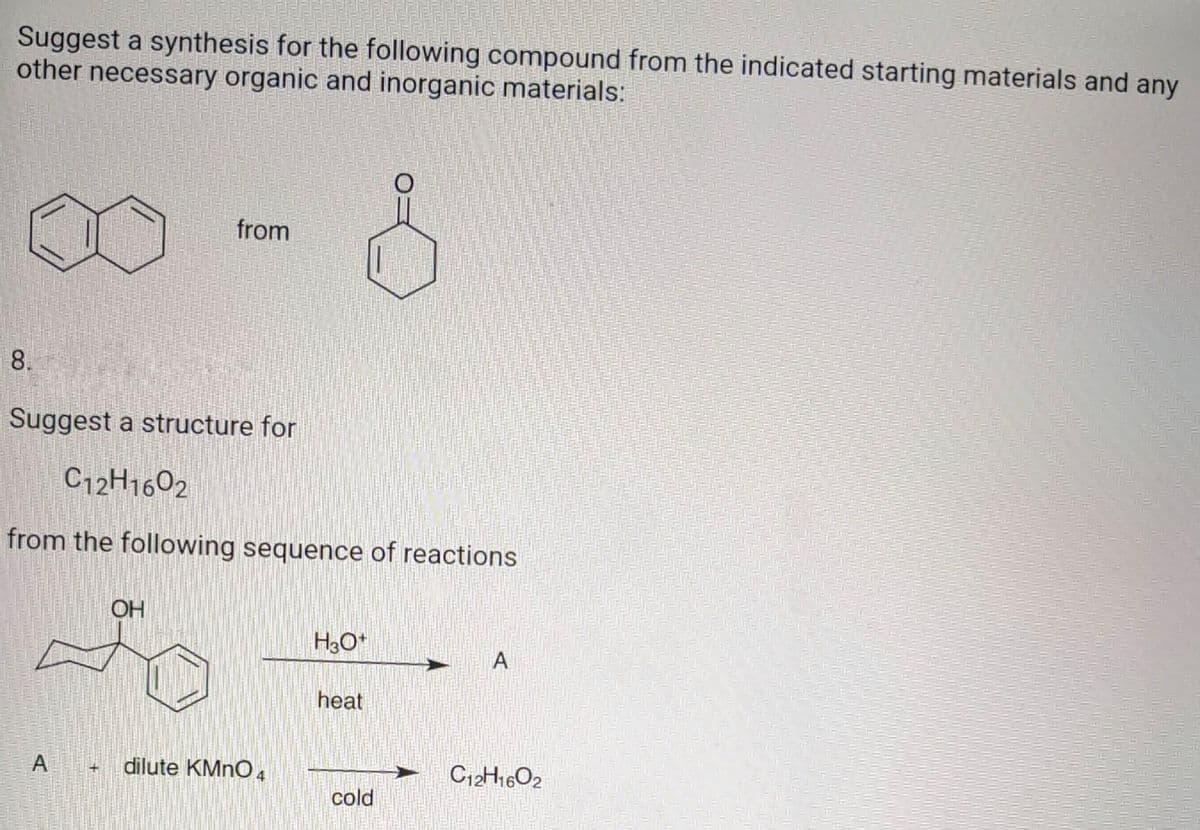
Suggest a synthesis for the following compound from the indicated starting materials and an other necessary organic and inorganic materials: from 8
Q: 6. Which statement(s) is/are true? I The solubility of most ionic compounds in water increases with…
A:
Q: GUIDE QUESTIONS: 1. What are other oils, besides coconut, that are used to make bath soaps? 2. Based…
A: (1) There are some other oils besides coconut oil - * Sun flower oil * Sweet almond oil * Olive oil…
Q: Suggest a synthesis for the following compound from the indicated starting materials and any other…
A: Since, you have multiple questions. So, I will solved first question for you. You can see the…
Q: Which of the following does have the lowest ionization energy? Z of Mg =12, Ca= 20, Sr =38, Ba =…
A:
Q: Aqueous hydrobromic acid (HBr) will react with solid sodium hydroxide (NaOH) to produce aqueous…
A:
Q: Given the following table of standard reduction potentials for a hypothetical element X under acidic…
A: First, we have to write all the balanced half-reactions for all of the given cell-notations. Also,…
Q: If 2.00 g of solid KO₂ is made to react with 1.00 g of CO₂ to produce solid K₂CO3 and oxygen gas,…
A:
Q: 12. A solution containing both Mn+2 and Cd+2 is saturated with H₂S gas, after which the…
A: According to bartleby guidelines we're supposed to answer only first question among these So please…
Q: Calculate the Mgo content of milk of magnesia, 12.32 g of which was dissolved in 50.00 mL of 1.0340…
A:
Q: AHvaforization = 2260 J/g your ne valadies gniwotlot de 2 by 01 Practice Problems 1. A copper pot…
A: 1) given, mass of a Copper pot (m) = 772 g Heat absorbed (Q) = 22.7 kJ = 22.7 × 1000 J = 22700 J…
Q: Assign R, S configurations to each indicated chirality center in the molecules below. A. OH B. COOH…
A: If the priority order is clockwise and hydrogen down then "R" If the priority order is clockwise and…
Q: What is the expected osmotic pressure of a 0.053 M solution of MgCl2 at 25.0 °C? Assume that the…
A: Answer is : a) 3.89 atm
Q: Sketch how the overlap between a 1s orbital and a 2p orbital directed towards it can be expected to…
A: The solution of the given question is
Q: Draw the missing products or reagents in the following multistep synthesis. Ignore any inorganic…
A: The reactions involves free radical halogenation of cyclopentane and then it's reaction with NaOH to…
Q: Which of the two resonance structures shown below would be a more significant contributor to the…
A: In given two resonance structures , first has positive charge on oxygen while in second positive…
Q: Question is based on the following definitions. Plants can propagate, or reproduce themselves, by…
A: This reproductive form resembles bulb most closely as in that too by dividing plant starts growing…
Q: 7. Name the following compounds: A) CoCl₂ Name: B) P3N5 Name: C) (NH4)3P Name: Name: D) GeO₂
A: Different chemical compounds in inorganic chemistry have different names depending on the metallic…
Q: A solution of 2.50 g of a compound having the empirical formula C6HsP in 25.0 g of benzene is…
A:
Q: A tripeptide contains lysine (R= -(CH₂)4NH2), phenylalanine (R= -CH₂C6H5) and glycine (R= -H). It…
A:
Q: How many distinct and unique isomers (excluding stereoisomers) can be drawn for…
A: The given coordination complex is Co(en)(H2O)(NO2)(F2).
Q: Standardization of a sodium hydroxide solution against potassium hydrogen phthalate (KHP) yielded…
A: The chemical reaction between KHP and NaOH is given by - KHP…
Q: CHARLE'S LAW PROBLEM SOLVING 1.) Under constant pressure condition, a sample of hydrogen gas…
A:
Q: Arrange the following in order of DECREASING molar entropy at 298 K. N2H4 Ar HF a. HF > N2H4> Ar b.…
A: As we know Greater the molar mass of the substance , more will be the molar entropy of the substance…
Q: Classify each reaction below as a(n): a. addition b. elimination C. substitution d. rearrangement…
A:
Q: Question # 2: a) Draw and name at least 5 structural isomers with the chemical formula C,H,O. The…
A: Structural isomers are the compounds which are having same chemical formula but different…
Q: Which of the compounds in each pair below, reacts faster by Sn 1? .**** Br A Br с O a. A and B O b.…
A: Correct answer is c (B and C)
Q: Question #1: Complete each reaction below. Name the organic reactant. Name and draw the product(s).…
A: These are reactions are markovnikov addition and Reduction reaction.
Q: 513/variants/434513/take/11/ Question 14 In the following reaction, what is being reduced and what…
A: Oxidation reduction reaction (Redox reaction) It can be defined as the chemical reaction in which…
Q: - How many MOLES of dioxygen difluoride are present in 2.51 grams of this compound? - How many GRAMS…
A:
Q: IDENTIFY DIFFERENT CHEMICAL REACTIONS AND ITS ADVANTAGES AND DISADVANTAGE
A: A Chemical Reaction is a process that occurs when two or more molecules collide with the right…
Q: 3 Cu +8HNO3 --> 3 Cu(NO3)2 + 2 NO + 4 H₂O In the above equation how many moles of water can be made…
A: Number of moles is calculated by dividing given mass with molecular mass.
Q: Provide IUPAC names for the following structures. H₂C Br CH3 a. Br Br H₂C b. Br Br
A: The IUPAC name of the given structures are:
Q: GRAMS of dioxygen difluoride are present in 3.52 moles
A: Given, Mass = 2.51 g Molar mass of O2F2 = 70 g/mol Moles = mass/molar mass = 2.51/70 = 0.0357…
Q: What is the minimum number of complete ice cubes at 0 °C needed to cool 500. grams of liquid water…
A: The energy lost or gained can be calculated by the multiplication of the mass of the compound (m),…
Q: Question 15 Which functional group is capable of participating in hydrogen bonding? O alkyne O…
A:
Q: Consider the following chemical reaction: CaO(s) + H2O(l) ----------> Ca(OH)2 (aq) if 1.00 g…
A: Given Mass of calcium oxide = 1.00 g Mass of water = 2.00 g
Q: Consider the reaction of 2-bromo-2-methylpropane with water, shown below, to answer the following…
A: In the second step the lone pair of water will be attack with the electron deficient planar…
Q: a. Which of the following mechanistic steps makes the most sense given the structures and charges…
A: A reaction will proceed in the forward direction if it form a stable intermediate or transition…
Q: Directions: Solve the following problems on a separate sheet of paper. Show your complete solutions.…
A: Non spontaneous reaction see below.
Q: For diatomic nitrogen (N₂), the highest occupied molecular orbital (HOMO) is lowest unoccupied…
A: HOMO - Highest occupied molecular orbitals LUMO- Lowest unoccupied molecular orbitals LUMO is…
Q: 185 mL. of a solution contains 1.55 g glucose. Calculate the molarity of solution. Glucose =…
A: Given, mass of glucose = 1.55 g Volume of the solution = 185 mL = 0.185 L molar mass of glucose =…
Q: How many MOLES of dioxygen difluoride are present in 2.51 grams of this compound? How many GRAMS of…
A:
Q: Select to Draw mCPBA Select to Draw OH PBr3 DMF H₂ Lindlar's catalyst HBr Select to Draw HCCNa THF…
A: Given reactions are : Predict all the products = ?
Q: Label each pair of stereoisomers below as: a. enantiomers b. diastereomers C. identical Place the…
A: The molecules with the same molecular formula but different structures are known as isomers. If the…
Q: Ammonia (NH3) gas and oxygen (O₂) gas react to form nitrogen (N₂) gas and water (H₂O) vapor. Suppose…
A:
Q: What amount of energy is required to change 20.0 g of an unknown substance from -15.0 °C to 7.0 °C?…
A: A substance exists in a solid as well as a liquid state at its melting point. The extra heat that is…
Q: 2. Sketch (as accurately as you can) steady state i vs E curves for solutions prepared from: a) 1 mM…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: A sample containing 1.00 g CaCO3 (100.09 g/mol) was allowed to react with 50.0 mL 0.720 M HCl. If…
A: The percentage yield is ratio of practical yield to that of theoretical yield, whole multiplied by…
Q: C OD. O B. OC. CH3 A Br CH₂ B AC... CH3 CH₂ Br H3C Br C CH₂ معاكم
A: For the most stable conformation, bulky group (Br) should be placed at equitorial position and…
Q: 6. Given 2CuO (s) → 2Cu (s) + O₂ (g) AH = 314.6 kJ a. Determine the heat of formation of CuO (s). b.…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Step by step
Solved in 2 steps with 1 images

- One of the products of petroleum refinery is naphtha where, benzene could beobtained via catalytic reforming of naphtha. The obtained benzene can potentiallyto react with Lewis acid to form new carbon-carbon bond. Propose the startingmaterial and stepwise mechanism to produce new chemical structure which consista formula molecule of C11H16.(a) Propose a reasonable synthesis for the formation of nonane from CH3CH2CH2I and any other organic/inorganic reagent. (b) Suggest three (3) different Grignard reactions leading to 2-phenyl-2-butanol.3. a. Write the complete reaction equation and give the name off all product (s) from the following reaction:i. bromination of phenolii. benzoic acid and ethylchloride in the presence of AlCl3b. Design the synthesis of two following compounds.i. m- chloropropiophenone from benzeneii. p-bromonitrobenzoic acid from toluene
- In an aqueous solution containing sodium bicarbonate, aniline reacts quickly withbromine to give 2,4,6-tribromoaniline. Nitration of aniline requires very strong conditions,however, and the yields (mostly m-nitroaniline) are poor.(a) What conditions are used for nitration, and what form of aniline is present under theseconditions?Stearolic acid, C18H32O2, yields stearic acid on catalytic hydrogenation and undergoes oxidative cleavage with ozone to yield nonanoic acid and nonanedioic acid. What is the structure of stearolic acid?Compound A has the molecular formula C14H25Br and was obtained by thereaction of sodium acetylide with 1,12-dibromododecane. On treatment ofcompound A with sodium amide, compound B (C14H24) was obtained. Ozonolysisof compound B gave the diacid HO2C(CH2)12CO2H. Catalytic hydrogenation ofcompound B over Lindlar palladium gave compound C (C14H26), andhydrogenation over platinum gave compound D (C14H28). Further, C yieldedO=CH(CH2)12CH=O on ozonolysis. Assign structures to compounds A through Dso as to be consistent with the observed transformations.
- Propose a synthesis of the anti-inflammatory drug Ibuprofen from benzene. Show all reagents and all intermediate structures. Assume that ortho and para isomers can be separatedA hydrocarbon (X), with the molecular formula: C8H14 is reduced in presence of sodium and liquid ammonia to give the only product (Y) with the molecular formula: C8H16. Compounds X and Y both resulting 2,5-dimethylhexane when treated with hydrogen and platinum catalyst (H2/Pt). As a result of the oxidative cleavage of compound Y (by using KMnO4 / H2SO4), a single carboxylic acid derivative with C4H8O2 molecular formula is formed. Again, as a result of the reaction of Y with perbenzoic acid, the chiral compound C8H14O is observed, but the reaction of compound Y with bromine gives the achiral C8H14Br2 as the product.a. Provide the structure of the hydrate of cyclopentanone. b. Provide the major organic product which results when pentanal is subjected to the following sequence of steps: 1. PhMgBr; 2. H3O+; 3. PCC.
- 1.Describe the ozonolysis of alkenes 2.one mole of a hydrocarbon(A) reacts with one mole of beomine giving a dibromo compound C5H10Br2.Substance A on treatment with cold dilute kMnO4 solution forms a compound C5C12O2(C5H12O2) on ozonolysis A,gives equimolar quantities of propanone and ethanol.Deduce the structure of substance A.Starting from indole, propose a synthesis of disubstituted indole E. More than one step is required. Outline reagents and conditions required to accomplish each step, identify key intermediates and discuss any point of selectivityBenzil reacts with 1,2-diaminobenzene to give a compound with molecular formua C20H14N2. Suggest a structure for this compound and write a reaction mechanism to show how it is formed. Do not give handwriting solution.

