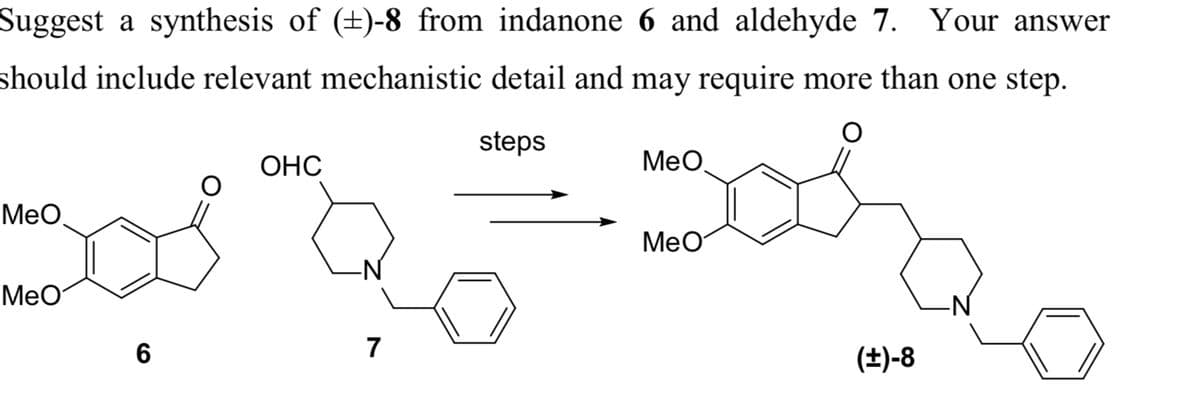
Suggest a synthesis of (±)-8 from indanone 6 and aldehyde 7. Your answer should include relevant mechanistic detail and may require more than one step. steps ಕ MeO MeO OHC 7 MeO MeO (+)-8
Q: At 37°C, the serine protease subtilisin has keat = 50 s¹ and KM = 1.4 x 10-4 M. It is proposed that…
A: On a Michaelis Menten plot, Km = [S] when vo = vmax/2. Km describes the enzyme's affinity for the…
Q: Which of the following cofactors is required for the conversion of succinate to fumarate in the citr…
A: Enzymes are proteins (ribozymes being the exception) that catalyse biochemical reactions.Some…
Q: Click the "draw structure" button to launch the drawing utility. Draw the structure of the…
A: Deoxyribonucleotide is a structure that is major component of DNA. It is made up of three parts.…
Q: Consider the role of Histidine in the Serine protease mechanism and sketch a plot showing he…
A: Enzymes are biological catalysts that increase rate of biochemical reactions.Serine proteases are…
Q: Site-specific recombination is often virus integration SARS-CoV-2 replication DNA repair pre-mRNA…
A: Site-specific recombination refers to a type of genetic recombination that occurs at specific target…
Q: Please answer as soon as possible!!*** Can you check if the answer below has correct information/if…
A: Body cells take in glucose from the blood with GLUTS. Some are insulin sensitive whereas some are…
Q: Why does isolated DNA appear stringy?
A: DNA is also known by the name Deoxyribonucleic acid. It comprises of the genetic instructions that…
Q: The COVID vaccines made by Pfizer and Moderna both utilize a new technology based on mRNA. d. What…
A: The COVID-19 pandemic has been mostly contained thanks to COVID vaccinations, which offer excellent…
Q: In Biochemistry there is a key aspect which is regulation. It affects all aspects of the cell life…
A: It is important to regulate reactions and metabolic pathways. This enables the body to choose which…
Q: 8. Use carbon oxidation number to help balance the following reaction with NADH and NAD. Clearly…
A: To balance the reaction with NADH and NAD, we make sure that the number of electrons transferred is…
Q: Upon exposure to glucagon, what is the phosphorylation state and activity of Triacylglycerol Lipase?…
A: A vital component of controlling blood sugar levels is the pancreatic hormone glucagon. It…
Q: 3. Below are abbreviated Haworth projections of 4 sugars. Label the sugars as a or ß. Which sugars…
A: The question is related to the D and L, classification of carbohydrates.D form: If the OH group of…
Q: 1. What happens to electrons in an oxidation reaction? Electrons are shared equally. Electrons are…
A: Note: since you have posted multiple questions we will solve first three questions for you as per…
Q: 10,000 2.000 66667 Draw Lineweaver-Burk plots for the behavior of an enzyme for which the following…
A: For a one-substrate enzyme-catalyzed reaction, the Michaelis-Menton equation shows the quantitative…
Q: Which statement accurately describes structure A in the figure above? B. H A. Н c=0 H-C-OH НО-C-H…
A: D and L designations of sugars are given on the basis of position of the hydroxyl group on the…
Q: 1. Label the ALPHA-Carbon, AMINO Group, CARBOXYL Group, and AMINO Side Chain of the following amino…
A: Amino acids are biomolecules that have a hydrogen, an amino group and a carboxyl group linked to the…
Q: A graph of 1/vo vs 1/[S] is shown below. The y intercept is 6.6 and the slope is 133.3. Determine…
A: We know that LB plot is a doublerms reciprocal plot which is made by plotting inverse of substrate…
Q: What ultimately powers the synthesis of ATP in cells? Group of answer choices Electrochemical…
A: The question is asking about the ultimate source of energy that powers the synthesis of ATP…
Q: The most commonly used biochemical reducing agent is: a) NADH b) NADPH c) FADH2 d) LiALH4…
A: The most commonly used biochemical reducing agent is NADH. NADH is a coenzyme that functions as a…
Q: Which statement concerning lipid ctions is O a. Steroid hormones act as messenger molecules and…
A: Lipids, hydrophobic organic molecules, serve vital roles in living organisms. Triacylglycerols store…
Q: Explain the mechanism of action of ACE inhibitors in managing hypertension
A: ACE inhibitors are Angiotensin-converting-enzyme inhibitors which are mainly used for the treating…
Q: A reaction with a free energy of hydrolysis can be coupled to the hydrolysis of ATP to ADP and Pi.
A: ATP stands for Adenosine Triphosphate. It is known as the energy currency of the cell. The…
Q: Give the number of the reactions(s) of the citric acid cycle that reduce FAD give the order number…
A: The citric acid cycle is the most active pathway in aerobic lifeforms that is responsible for…
Q: Please give me all answer 1 . Which one of the given amino acids has a sulfur atom? A. cysteine B.…
A: Amino Acids:Amino acids are the building blocks of proteins, essential molecules in living…
Q: The concentration of hydrolyzed nitrocefin at each time point for an experiment is given below.…
A: Nitrocefin is a chromogenic cephalosporin substrate. This substrate is used to detect the presence…
Q: CH3CO3H
A: The given reaction is a reaction of ketone and per acid. The product of this reaction is an ester.…
Q: 3. Below is the structure of a nucleotide triphosphate. of of of O ATP R-O- R-O O R-O O O O O +|| O…
A: Nucleotides are organic molecules that form the basic building blocks of nucleic acids (DNA and…
Q: ng at the figure above, which statement is true? B. H H H-C-OH HO-C-H H-C-OH H-C-OH CH₂OH =0 HO-C-H…
A: Stereoisomers of sugars are broadly divided into enantiomers and diastereomers. 2 stereoisomeric…
Q: The tyrosine absorption spectrum shows two peaks, one at 225 nm and one at 272 nm. What is the…
A:
Q: the reaction: peptide (20 residues) + H₂O + chymotrypsin → peptide (8 residues) + peptide (12…
A: Chymotrypsin is a proteolytic enzyme, meaning it catalyzes the hydrolysis of peptide bonds in…
Q: How to draw the haworth projectioj of common cyvlic form of monosaccharide
A: Haworth projection is a way of representing the structure of monosacharides.Monosacharides are the…
Q: 19. Endorphins are polypeptides that reduce pain. What is the amino acid order for the following…
A: The body produces endorphins, which are neurotransmitters that can provide sensations of pleasure or…
Q: Which of these is not a macromolecule? a) Ala b) protein c) polysaccharide d) nucleic acid The…
A: The objective of this question is to identify which of the given options is not a macromolecule.…
Q: Velocity, activity units/mg protein 51 3 2 0 5 15 20 Aspartate concentration, mM - 10 Control With…
A: Enzymes that are under allosteric control can be categorized into two groups;K-series enzymes :…
Q: Please answer the following answer: Ethanol metabolism has been part of the human diet for…
A: The question is asking about the effects of high concentration of NADH, a product of ethanol…
Q: 8.) Monosaccharides - 2 A. Circle the stereocenters in this monosaccharide structure. B. Identify…
A: The given question relates to the stereochemistry of carbohydrates.Stereocenter: The carbon center…
Q: What is the net yield of ATP when each of the substrates is completely oxidized to CO₂ by a…
A: We know through electron transport chain cycle ATP is generated from NADH and FADH2 equivalent as…
Q: Your roommate works in a research lab focusing on improving livestock feed. She you she's working…
A: There are four classes of biological macromolecules: nucleic acids, proteins, lipids and…
Q: Binding of acetylcholine to postsynaptic membranes increases the conductance of Na+ into the cell.…
A: The question is asking us to identify the type of transport mechanism that is involved when…
Q: As electrons fall from one complex to another in the Electron Transport Chain, the energy they…
A: Oxidation of 1 molecule of glucose can produce 10 NADH (6 from acetyl CoA in the TCA cycle + 2 from…
Q: Which ligand-binding curve in the figure below would be consistent with the protein exhibiting a Ka…
A: Consider the interaction between a protein (P) and ligand (L) as shown below.Here, Ka is the…
Q: All of the following statements about the B-oxidation pathway are true except: The pathway is…
A: In metabolism, β-oxidation is the catabolic process by which fatty acid molecules are broken down.…
Q: Which of the following statements are true? (This is a multi- select question, select all that…
A: There are four classes of biological macromolecules: nucleic acid, proteins, lipids and…
Q: Categorize each of the following carbohydrates as an aldopentose, aldohexose, or ketohexose. Drag…
A: Monosaccharides can be divided into aldoses and ketoses based on the functional group in…
Q: Which of the following regarding DNA replication is FALSE. Replication of DNA is highly regulated.…
A: There are four classes of biological macromolecules: Nucleic acids, proteins, lipids and…
Q: Enalaprilat is a competitive inhibitor of the angiotensin-converting enzyme (ACE), which cleaves the…
A: Here, ACE is the enzyme, angiotensin I is the substrate and enalaprilat is the inhibitor.Given…
Q: The original DNA sequence was CCGGAATT. It was replicated. Which of these newly replicated DNA…
A: DNA is deoxy ribo nucleic acid which copies itself by a process called replication. This helps in…
Q: What does an enzyme change relative to an uncatalyzed reaction? a) The rate of the reaction. b) The…
A: Enzymes are biological catalysts. All enzymes are proteins except some RNA molecules that also show…
Q: Draw the structures of pyruvate, Acetyl-CoA, alanine, oxaloacetate, and glyceraldehyde-3-phosphate…
A: Pyruvate is the end product and glyceraldehyde-3-phosphate is the intermediate of the glycolytic…
Q: Fructose-2,6-bisphosphate is an activator of and an inhibitor of O phosphofructokinase; hexokinase O…
A: GlycolysisIt is a process of conversion of glucose into pyruvate and generates energy in the form of…
Provide reaction mechanism
Step by step
Solved in 3 steps with 1 images

- Show the complete and detailed reaction mechanism (Curtius Rearrangement) of carboxylic acid 4 that resulted in the formation of isocyanate 5, which, upon electrocyclization furnished pyrrolo[2,3-c]quinoline 6. Intermediate 6 was subsequently transformed into triflate 7, and the total syntheses of marinoquinolines 3a, 3b, and 3e were accomplished by intro- ducing different substituents at the C4 position. (show movement of electrons, charges, arrows, reagents, conditions, biproducts etc. and provide short explanation)Electrophilic nitration of benzoic acid gives almost exclusively 3-nitrobenzoic acid. By drawing the appropriate resonance forms of the intermediate cations resulting from attack of [NO2]+, explain this result.1..Suggest mechanisms for the following reactions:
- Please describe the difference in the outcome of treating a terminal alkyne with HgSO4, H2SO4, H2O and with 9-BBN, followed by H2O2 and NaOH.Following is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?8. The preparation of the sex pheromone of the boll worm moth, (E)-9,11-dodecadien-1-yp acetate from compound A has been described. Suggest suitable reagents for each step in sequence.
- When a mixture of ketone 11 and \alpha, \beta - unsaturated carbonyl 12 are exposed to substoichiometric amounts of NaOMe, product B is obtained selectively. Subsequent treatment of B with excess NaOMe leads to the exclusive formation of tricyclic product 13. (1) Suggest a structure for B and provide a reason for its selective formation. (ii) Provide a reaction mechanism for the formation of 13, which accounts for its selective formation.I need help to find the mechansim for Preparation of Cyclohexanone by Na2CrO3/H2SO4 and explain the mechanisms step by step so I can understand it pleaseWhen 2,2-dibromo-1-phenylpropane is heated overnight with sodium amide at 150 °C, the major product (after addition of water) is a different foul-smelling compound of formula C9H8. Propose a structure for this product, and give a mechanism to account for its formation.
- Propose a mechanism for the reaction of aniline with ethyl acetate to give acetanilide.What is the leaving group in your proposed mechanism? Would this be a suitableleaving group for an SN2 reaction?9. Product of the reaction of nitrobenzene with sulfuric acid in the presence of oleum at 60 °C:a) o-nitro benzenesulfonic acidb) m-nitro benzenesulfonic acidc) p-nitro benzenesulfonic acidd) None of the above 10. Due to their ability to move through ducts or pipes, they are generally called fluids:a) Solidsb) Solid mixturesc) Gasesd) Liquids and gasesReaction of Aldehydes and Ketones with Organometallics: Grignard and Lithium For the following reactions: 1) Illustrate the reaction for the formation of the organometallic 2) Write the product of the reaction 3) Illustrate the mechanism of the reaction with arrow movement.

