Q: when necessary. a) b) Br2. H20 HCI d) H2 Ni MCPBA 1) ВН; THF 2) H2O2 NaOH f) H2SO4, HgSO4 H3O*
A:
Q: An organic acid can be represented by: ‖ CH3 – CH2 – C – H…
A: A multiple choice question based on functional group in organic compounds, which is to be…
Q: 9. NAHSO3 CH3 - C- CH3 19)_ 10. (10a) NaOH → HC-O-Na + _(10b)_
A: These are the oragnic reactions which include organic compounds that breaks or combines to give a…
Q: u. CH3 hv H3C- V. H3C. N. Ph C(CH3)3 H3C HO, W. CH3 МСРВА CF3
A: Since you have asked multiple questions, we will solve first three questions for you. If you want…
Q: I CH2(CO2Et)2 R- .COOH II RCH(CO,Et)2 R. „COOH III ACCH,CO,Et R. COCH; IV RCH,COCH; R. COOH R. COOH…
A: Iodoform test is used to detect the presence of carbonyl group.
Q: 9. NAHSO: CH: - C- CH3 19). 10. L(10a) + + NaOH HC-O-Na + _(10b)_
A: Acetone reacts with sodium bisulphite to form acetone bisulfitethis is addition reaction.
Q: Draw the organic products formed in each reaction.
A:
Q: Fill in the blank boxes below. 1. 1. 2. 9. 1. BH THF 2. HO, HO2 RO H Pd/C HCI HO 3. Cl2 CH-C HCI 7.…
A: The explanation of the organic reactions are given below in detail
Q: 1. 2. 3. 4. 5. HC1 + CH3I -CH3 + HONO N-H + a N- -H NHCH3 + HONO
A: Here we have to write the major product formed in the following given reactions.
Q: Question attached
A: Organic reactions are reactions of organic molecules. They undergo different types of reactions like…
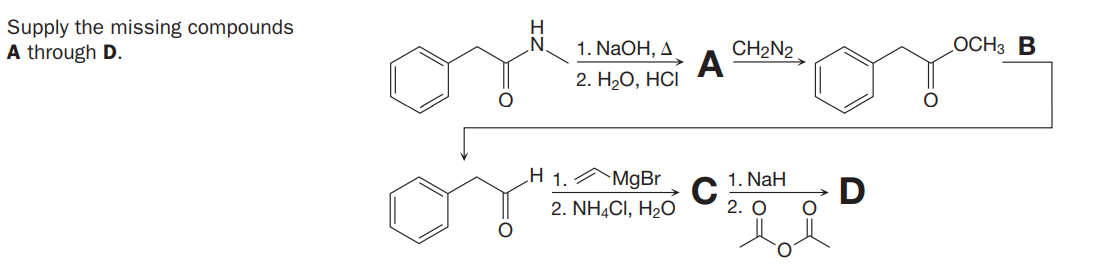
Q: Supply the missing compounds A through G. 1. KMNO4, КОН, Н2О CH,N2 1. BH3-THF A В C 2. H2О, НCI 1.…
A: The reaction of converting alkenes to an alcohol by the addition of BH3.THF in a base and in…
Q: HOCH2CH2OH H* Br2 (excess) HO. 7. NaOCH3 CH3OH C6H5 5. 6.
A:
Q: CH₂ Br a. 98-15 and 2 4 3 Br CH₂ C1- C2- C3- C4- Related- b. Me 1 Me OH and Et OH C1- C2- Related-…
A: Here we have to determine R and S configurations of chiral centres and relationships between the…
Q: NO2 Cle gas NCS. NO aq. NaOH NO2 "NH NH SCN Br NH2 (4) (2) (3) ag. NASH NCNHCOOCH, NH2 HC CH3 aq.…
A: Ans
Q: CHEM 111 Nomenclature Lab 2C 2)b. K3ASO4 d. Caз(РОa)2 f. Na2SiO3 h. K2Cr2O7 j. KMNO4 1. L12SO4 n.…
A: 2) b. K3AsO4 - Potassium Arsenate
Q: 요 он + + H₂N -N- H O_H "NH₂ HCI e troce acid >
A:
Q: NaBHa, Oн a) Hg(OAc)2 p-TSCI b) KCN c) ► d) pyridine
A: See synthetic route below.
Q: For each reaction, identify the carbonyl AND the alkyl halide starting materials that form the…
A:
Q: Add more NH3 what will happen to H2?
A: We have to tell the effect of adding more NH3 what will happen to H2
Q: Name the following:
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: Name the family to which each organic compound belongs. The first answer has been filled in for you.…
A:
Q: Name the following:
A: The naming of the compound is done according to IUPAC rules for nomenclature. IUPAC naming is…
Q: Compound A Compound B HO Он NH2 HO он H3C. CH3
A: The given compounds: 1(A) 1(B) 2 (A) 2 (B) 3(A) 3(B) These compounds have to be identified.
Q: 4. Pb + H3(PO4) → H2 +_ Pb3(PO4)2 -- 5. Li3N + NH4 (NO3) - LINO3 + (NH4)3N - 6. HBr + Al(OH)3 → H20…
A:
Q: Supply the missing compounds A through D. 1. I2, NaOH 1. LIAIH4, THE PBr3 A 2. H.О, НC В 2. H20,…
A: Reagents are the chemical species that can be used to carry out a specific reaction on the provided…
Q: CHOR Cl2 FeCl HIA NH2 H2 Pt HCI O P(O)s Benzene u) + CN 1) NAOCH/HOCH, OCH3 2)HHO HBr OH H,CrO4 1)…
A:
Q: 1. HNMe2,pyridine 2.LİAIH, 3. Н,О H,c `Cl 2-7 1.SOCI, 2. NH, 3. NAOH, Br 2-8 1. NAN3 2. LIAIH, 3.…
A: LiAlH4 reduces amide to amine SOCl2 converts COOH to COCl NaOH, Br2 convert CONH2 to -NH2
Q: Multi-step synthesis
A: The multistep reactions can be shown as :
Q: OH IH HO Нeat TSOH (b) (а) ОH SOCI HO H2SO4. (d) (с) HBr OH PBr3 (f) OH OH POCI, (g) Руridine CH3…
A: a. Here, alcohol gets converted into OTs. b. Here, Propanol on reaction with HI gives propyl…
Q: Alkyl halides 1.1 CH3 CI CH,OH A CH3 K' OC(CH,), H3C Figure 1 1.2 CH3 Na OCH,CH, C Br H3C Figure 2
A: Alkyl halides are highly reactive compounds due to the presence of polar carbon-halogen bond in…
Q: _____ H3PO4 + _____ KOH ----> _____ K3PO4 + _____ H2O
A: Complete the reaction--- _____ H3PO4 + _____ KOH ----> _____ K3PO4 +…
Q: H 138 NH₂ 121 H 140 i) (MeO)2SO2. ii) HCHO, HCI, DMF 189, 67% HN dioxane/H₂O, 60-80 °C 62-86%…
A: The first step of the synthesis is the condensation reaction between phenylhydrazine (138) and…
Q: NaOH H;C--o-CH3 H;C- Na но-сн, + H20
A: Addition of nucleophile Loss of leaving group Proton transfer
Q: What is the functional class in each molecule (alcohol, amine, thiol, etc.). Write your answer below…
A: Functional group is a group of collection of element which shows specific chemical and physical…
Q: MIG HBr, ROOR BAT Pool of Choices CH3 c JOH BIG cold dilute neutral KMnO4 CH3 Br IOH Br₂, H₂O PIG…
A:
Q: Dob OH 1. LIAIH, (excess) 2. H₂O NaBH₁ H₂O HC1 Mg ether 1) MgBr, ether 2) H*, H₂O NH, 1) LIAIH, 2)…
A:
Q: 9. NAHSO3 CH3 – C- CH3 19). 10. _(10a)_ NaOH → HC-O-Na + _(10b)__
A:
Q: d) e) Br 1- 2- Ph +H' Br 3- H¹, H₂O NaOEt EtOH 1. PhMgBr (excès) 2. H3O* 1-03, CH₂Cl2, -78 °C…
A:
Q: H₂C. CH,(CO,Et)2 + 8 + Ph EtO H acetone O NaOH, H₂O Δ .CO₂Et OEt NaOEt 1 NaOEt, EtOH 2 H3O+
A:
Q: 6 S Cr₂O3 Base RX 4 KNO3 H₂SO4 carbonyl punodwoo HCI/Zn 2 3
A: The main substrate we have is 1H-indole-2,3-dione, commonly known as isatin. The structure of isatin…
Q: Rank the following compounds from least [1] to most [4] acidic. OH OH NH2 NH2 II III IV © GMU 2020 v…
A: Acidity order of the following compounds.
Q: (i) (1) BH3 THF B (ii) H2O/HO (ii) Br SH Br2 (i) POCI, HO E CH2CH3 Pyridine (iv) (i) R2BH (ii) H2O2,…
A: Four Organic chemistry problems, which are to be accomplished.
Q: с) HBr d) H* NaOCH3 HOCH3 HOCH3
A:
Q: Но HN NH H2N-
A: RCOOR' = ester ROR' = ether RNH2 = primary amine R2NH = 2°/ secondary amine RCOOH = carboxylic…
Q: K H NaOEt, E1OH NC. CN NaOEt, EtOH H H. M NaOEt, EtOH
A: 1) Reaction of an active methylene compound with carbonyl compounds is called Knoevangel…
Q: 1) OH X Br out 1) Mg, ether 3) H₂O (CH3)3COOH Ti[OCH(CH3)2]4 (+) DET LOCH₂CH3 1. CH₂ONa 2. H₂O 1) xs…
A: The reaction has been done by considering that nucleophile attacks in less hindered site.
Q: OTBS PCC F) DCM, 25 °C 1) Hg(OAc)2. THF/H20 2) NABH4. Na OH TIPSOTI, pyridine H) DCM, 25 °C
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Need help on my last problem (N2+3H2—->2NH3)Excessive nutrients dumped into waterways can create “algae blooms” that rapidly consume all of the dissolved O2 and trigger production of enough H2S to create “dead zones” like the ones that exist off the coast of Denmark and in the Gulf of Mexico immediately south of the Mississippi River delta. a. Since Group 16 element substitution is a recurring theme in that Group’s chemistry –e.g., oxygen substituting for Se and Te in the production of H2Se and H2Te, respectively –maybe molecular oxygen (O2) could be bubbled into these dead zones and react with the H2S.Write out a balanced chemical equation for the reaction between H2S and O2 to form elemental sulfur (S8) as a product that would precipitate out. b. Using bond dissociation energies, calculate if this reaction would be thermodynamically favorable. c. If your answer to part (b) is “yes”, do you think that simply bubbling O2 into these waters would work? Justify your answer.Consider following reaction: HgO (s) -> Hg(l) + ½ O2 (g) Delta H = +90.7 kj/mol. What quantity of heat in kj/mol is required to produce one mole HgO? Write your answer without units. Given the following data 2ClF(g) + O2(g) --> Cl2O(g) + F2O (g) Delta H= 167.4 kJ I 2ClF3(g) + 2O2(g) --> Cl2O(g) + 3F2 O (g) Delta H= 341.4 kJ II 2 F2(g) + O2(g) ---> 2F2O (g) Delta H= -43.4 kJ III Calculate the delta H in kJ for below reaction: ClF(g) + F2(g) ---> ClF3(g)
- Tu Re-order each list in the table below, if necessary, so that the atoms or lons in it are listed in order of decreasing size. Please don't provide the handwriting solutionRedraw the table below and then fill in missing information for the particular species in each of the column. Can you help me, please? I struggled with my homework.Help with Part A B and C: Ksp Values: Ionic CompoundFormulaKsp Aluminum hydroxideAl(OH)31.8×10–5 Aluminum phosphateAlPO46.3×10–19 Barium carbonateBaCO35.1×10–9 Barium chromateBaCrO41.2×10–10 Barium fluorideBaF21.0×10–6 Barium hydroxideBa(OH)25×10–3 Barium sulfateBaSO41.1×10–10 Barium sulfiteBaSO38×10–7 Barium thiosulfateBaS2O31.6×10–6 Bismuthyl chlorideBiOCl1.8×10–31 Bismuthyl hydroxideBiOOH4×10–10 Cadmium carbonateCdCO35.2×10–12 Cadmium hydroxideCd(OH)22.5×10–14 Cadmium oxalateCdC2O41.5×10–8 Cadmium sulfide*CdS8×10–28 Calcium carbonateCaCO32.8×10–9 Calcium chromateCaCrO47.1×10–4 Calcium fluorideCaF25.3×10–9 Calcium hydrogen phosphateCaHPO41×10–7 Calcium hydroxideCa(OH)25.5×10–6 Calcium oxalateCaC2O42.7×10–9 Calcium phosphateCa3(PO4)22.0×10–29 Calcium sulfateCaSO49.1×10–6 Calcium sulfiteCaSO36.8×10–8 Chromium(II) hydroxideCr(OH)22×10–16 Chromium(III) hydroxideCr(OH)36.3×10–31 Cobalt(II) carbonateCoCO31.4×10–13 Cobalt(II) hydroxideCo(OH)21.6×10–15 Cobalt(III) hydroxideCo(OH)31.6×10–44…
- compound: chemical trioxide please can u check if the answers are correctGroup A: solution conductivity (MS) NaCl 6594 CaCl2 13188 AlCl3 16311 Group B: solution conductivity (MS) HCl 21809 Hc2H3O2 639 H3PO4 8805 For H3PO4, does the subscript "3" of hydrogen in this formula seem to result in additional ions insolution as it did for AlCl3in Group A? Explain.Draw an octet-rule Lewis structure for POCl3: Total count of valence e¯’s: a) in the formula = __ b) as dots and dashes in your O.R.-Lewis structure = _____ Bond orders: P–O _______ P–Cl _______ (like triple in N2, single in H2O …) Count e¯-groups around P: _____ --> e¯-group shape around P is ________________ Molecular shape is _______________ The molecule is ? not polar (mark one) Oxidation numbers: O ____ Cl ____ P ____

