Q: 5- Addition H2 to butyne give 6- Addition HBr to 3-hexyne give
A: Given, 5. Addition H2 to butyne give 6. Addition HBr to 3-hexyne give
Q: Consider the reaction of 2-bromo-2-methylpropane with water, shown below, to answer the following…
A: In the second step the lone pair of water will be attack with the electron deficient planar…
Q: 2-ethyl-3-hexyne 4-chloro-2-pentyne 4-heptyne QUESTION 2 In the addition of hydrogen bromide to…
A:
Q: Predict the major products for the reaction of 1-methyl-1,2-epoxycyclopentane with H2SO4 in ethanol
A: The reaction of epoxide with acid and bases proceeds through the mechanisms of Sn1 and Sn2. The…
Q: For the reaction, give the expected substitution product: a) 1-iodo-1-methylcyclohexane +…
A:
Q: The reaction of 3-methylene-1-cyclohexene and HBr yields the four products shown in the attachment.…
A: At high temperature, thermodynamically more stable products are formed i.e. alkenes which more…
Q: what are the reagents/conditions that would transform cyclohexanol to 1-methylcyclohexene.?
A:
Q: How many monochlorination products are possible for the alkane shown below?
A:
Q: Identify the type of reaction of the following: Conversion of trans-1-chloro-2-methylcyclopentane to…
A: The given reaction is Conversion of trans-1-chloro-2-methylcyclopentane to 1-methylcyclopentene. The…
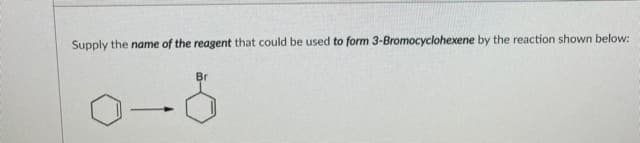
Q: Supply the name of the reagent that could be used to form 3-Bromocyclohexene by the reaction shown…
A:
Q: Reaction of propene (above) with HBr might, in principle, lead to a mixture of two alkyl bromide…
A:
Q: 2. Starting from benzene, synthesize the following molecules. You may use other organic and…
A: The given compounds are represented as follows:
Q: A B D ZO
A: The alkyl halide is a molecule in which the hydrogen atom of one carbon atom is replaced by the…
Q: Which one of the following alkenes is the Zaitsev product formed by the reaction of hydroxide with…
A: Elimination reactions usually produce the more highly substituted alkene, called the Zaitsev…
Q: Provide the structure of the major organic product: H,C. ...CI CH3CH2OH
A: In the above reaction, the alkyl halide reactant given is a secondary alky halide i.e the carbon…
Q: 3. Given the structure below; -ÖH H 3.1. 3.2. 3.3. OH =0: Use arrows to show the mechanism that…
A:
Q: If 1-bromopentane is heated in acetone containing NaOH, what is the alkane produced? Draw and…
A: Here 1- bromopentane is heated in acetone containing NaOH. Here acetone is a polar aprotic solvent ,…
Q: Provide the structures for each compound name given below, including notation for stereochemistry…
A: The compounds given are,
Q: Do the following conversions in not more than two steps:(i) Benzoic acid to benzaldehyde(ii) Ethyl…
A:
Q: H2SO4 + H H.
A: Hydration of alkene: Alkene gives hydration reactions in presence of an acid catalyst. In this…
Q: In the reaction of 3-hexene with HBr, which of the products is expected to be formed in a larger…
A: Alkenes are the compounds which contain one or more double bonds. The starting material is 3-hexene.…
Q: When cyclo-pentene interacts with hydrogen, it gives
A: Since we know that when an alkene react with hydrogen reduction of alkene occurs to form alkane.
Q: We are synthesizing an Alcohol by Hydrating an Alkene, We are using hex-1-ene and sulfuric acid.…
A: The reactants given are hex-1-ene and sulfuric acid.
Q: CH=C-CH2–CH2-CH3 H2 + Pt, Pd, or Ni A) + HBr B)
A: Answer: This reaction is based on addition reactions of alkynes and alkenes.
Q: If bromocyclohexane were converted into cyclohexene, what kind of reaction would that be?
A: One organic compound can be converted to another compound with the help of various reagents. This…
Q: Refer to the figure below (synthesis of hexylbenzene from styrene). Which of the given choices…
A: The detailed solution of your question given below-- For this synthetis we need to four more carbons…
Q: Explain this ? "Organic synthesis is the systematic preparation of a compound from areadily…
A: Introduction: In an organic synthesis, generally a small compound is taken and step by step after…
Q: Oxidation of a primary alcohol with pyridinium chlorochromate ( PCC, C5H6NCrO3Cl) would yield what?
A:
Q: 9. Complete the following halogenation reactions for benzene. Draw and name the product. a) b) + Br₂…
A:
Q: What would be the major product of the reaction between 3-methyl-1-butene and HCI?
A:
Q: Provide the structure of the major organic H; Ca ...C/ CH3CH2ONA
A: Nucleophilic substitution biomolecular reaction (SN2):The bimolecular nucleophilic substitution…
Q: If you react 1-hexene with hydrochloric acid with peroxides, what product will you produce? Write…
A:
Q: How would you synthesize the following compounds from butanoic acid using reagents from the table?…
A: In a chemical reaction, the substrate reacts with a reagent that is present to start the chemical…
Q: Two substitution products result from the reaction between 3-chloro-3-methyl-1- butene with sodium…
A:
Q: How can we convert benzene to m-chloronitrobenzene? Chlorination then nitration Nitration then…
A:
Q: Which of the following reagents would be used as a chemical test for the presence of alkenes and…
A: This is the concept of Electrophilic and Nucleophilic addition reaction.
Q: Illustrate the complete synthesis of 1,3,5-tribromobenzene from benzene ?
A: A synthesis is the manufacture of chemical compounds by the reaction from simpler materials or…
Q: ) begin with 1-bromopentane (shown), and make pentane
A:
Q: Explain why hexan-2-ol is formed as the major product when 1-hexene undergoes acid catalyzed…
A: Acid-catalysed Hydration of Alkenes by the addition of water molecule and strong acid,forms alcohol.…
Q: Predict the major products formed when 2- methyl-1-butene reacts with: HCl. Show the reaction…
A:
Q: (B). Write the chemical reactions of 2-chloro-2-methylbutane with the following two sets of…
A:
Q: CH HCHS 2. NAEH,
A:
Q: 9. The addition of a water molecule to 3,4-dimethylcyclohexene yields two possible isomers. Sketch…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Predict the products of the following reactions. 1-(bromomethyl)-2-methylcyclopentene, heated in…
A: The product of the following reaction has to be predicted. 1-(bromomethyl)-2-methylcyclopentene,…
Q: the addition of HI on cis-2-butene will yield which compound?
A:
Q: Provide the product(s) for the reaction of benzene with nitric acid and sulfuric acid. If more than…
A: When benzene is treated with Nitric acid and sulphuric acid then only Nitrobenzene is form as a…
Q: The mechanism of dehydration of a secondary alcohol to give an alkene can involve the formation of…
A:
Q: 1) 3- Methyl-2-butanol will react with H2SO4 to give two isomeric alkenes. Write suitable reaction…
A: Given Reactant = 3 methyl 2 butanol and H2SO4 Major Product = IUPAC name of each product = ?
name compounds
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Illustrate the Reaction of a Radical X• with a C=C ?The following radical-initiated reaction with HBr produces a formal Anti-Markovnikov's rule product. Provide a curved arrow mechanism, making sure to use single headed arrows and drawing all intermediates. Only initiation and propagation steps are needed. Termination does not need to be included. Part of the initiation step is provide for use to generate an OH radical.If you want to disrupt the ability of radicals to generate more of themselves, particularly in scenarios where they could be harmful like auto-oxidation, you need to find a suitable scavenger reagent. Such materials, known as antioxidants, succeed when they can lead to a new, more stable radical species that terminates the chain without reacting, or by further consumption of reactive radicals to generate additional non-radical species. Which species(s) is or are radical-scavenging reagent(s)? * I just II, only III, only IV, only I and II, only II and III, only III and IV, only
- In theory, there are only three inequivalent hydrogens in this molecule that could be substituted by Br in a free radical bromination – circle them. Put an asterisk to mark the one most likely to be substituted first.i wonder which number is the most reactive and the least reactive in sn2 reaction? can you rank them?Could someone help me with ranking these with how reactive they would be with an SN2 reaction? Thanks :)
- Draw the most stable radical that can result from cleavage of a C–H bond in attached molecule.What are ALL of the products when adding hexa-1,3,5-triene and bromine in CCl4? I know how to get the major product (both bromines next to each other due to epoxide) but don't know how to get to the other major and the minor product?Please help me with the organic chemistry problem below: Consider the reaction below: (Check the attached image) (it is between Furan and maleic anhydride, a DIels-Alder reaction) a) Will this reaction for an endo product (with a melting point of 80-81 degrees) or the exo product (with a melitng point of 114 degrees)? b) Carefully explain why the product must have been formed the way it did (exo or endo). c) Provide a mechanism for this reaction.
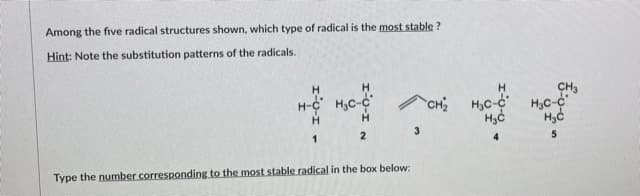
- From the given structure, indicate the most stable radical.Radical allylic bromination is also something that although I understand resonance structures I can't quite grasp how to approach problems like this. Thank you in advance for the help this site has been a lifesaver for my ochem understanding.In the presence of a radical initiator (Z•), tributyltin hydride (R3SnH, R = CH3CH2CH2CH2) reduces alkyl halides to alkanes: R′X + R3SnH → R′H + R3SnX. The mechanism consists of a radical chain process with an intermediate tin radical: This reaction has been employed in many radical cyclization reactions. Draw a stepwise mechanism for the following reaction.

