suppose 431. mIL of isopropanol and 1.42 L of water are mixed, and the final solution has a volume of 1.83 L. Note that the density of isopropanol is 0.79 g/mL. Calculate the volume percent of isopropanol in this solution. Be sure your answer has the right number of significant digits. %(v/») 1 Don't Know Submit 02022 McGraw Hill LLC AIl Rights Reserved Terms of Use Privacy Contr Ace 777 Mostly cloudy DELL
suppose 431. mIL of isopropanol and 1.42 L of water are mixed, and the final solution has a volume of 1.83 L. Note that the density of isopropanol is 0.79 g/mL. Calculate the volume percent of isopropanol in this solution. Be sure your answer has the right number of significant digits. %(v/») 1 Don't Know Submit 02022 McGraw Hill LLC AIl Rights Reserved Terms of Use Privacy Contr Ace 777 Mostly cloudy DELL
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter16: Solutions
Section: Chapter Questions
Problem 14E
Related questions
Question
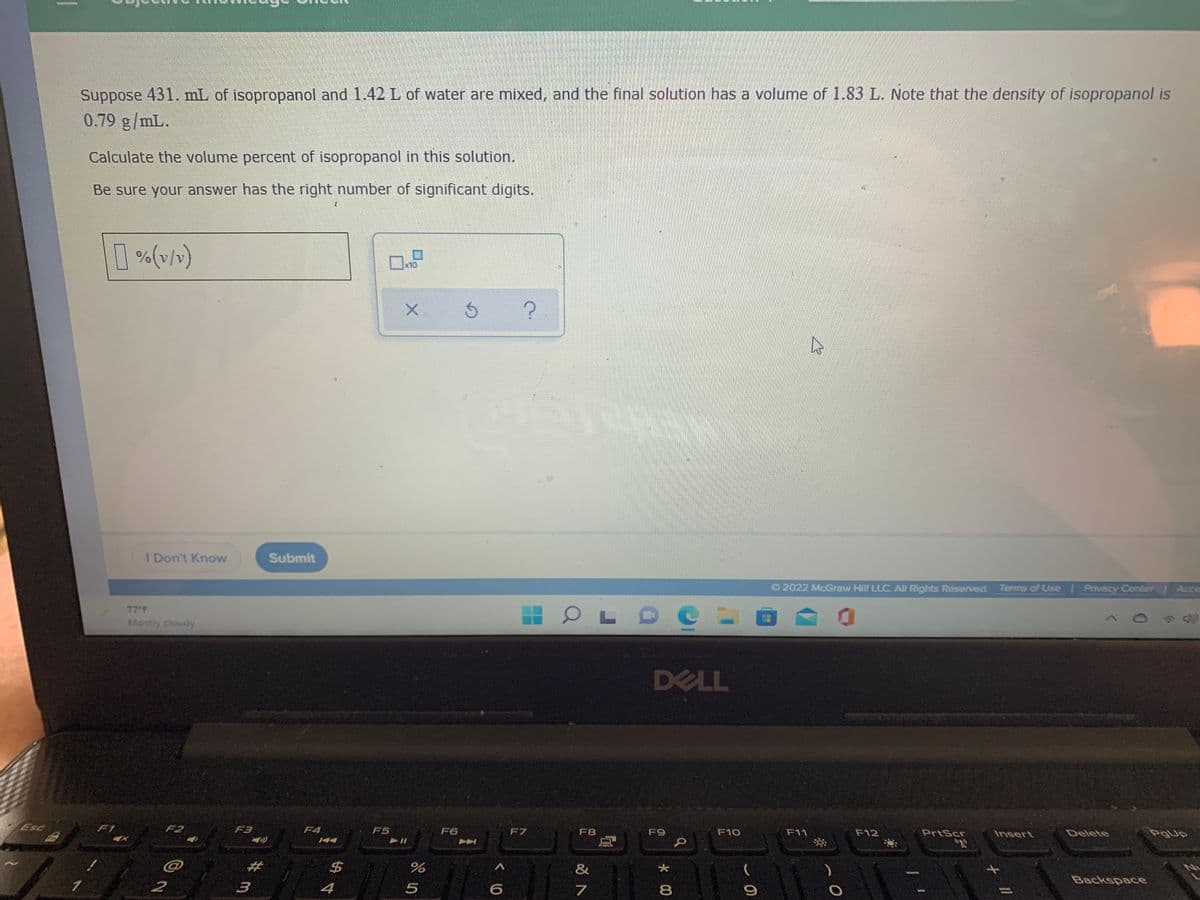
Transcribed Image Text:Suppose 431. mL of isopropanol and 1.42 L of water are mixed, and the final solution has a volume of 1.83 L. Note that the density of isopropanol is
0.79 g/mL.
Calculate the volume percent of isopropanol in this solution.
Be sure your answer has the right number of significant digits.
[ %(v/)
x10
I Don't Know
Submit
O2022 McGraw Hill LLC, All Rights Reserved. Terms of Use Privecy Center Acce
77 F
Mostly cloudy
DELL
Esc
F1
F2
F3
F4
F5
F6
F7
F8
F9
F10
F11
F12
PrtScr
Insert
Delete
PgUp
2#
24
&
Backspace
2
4
7
8.
II
* 00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning