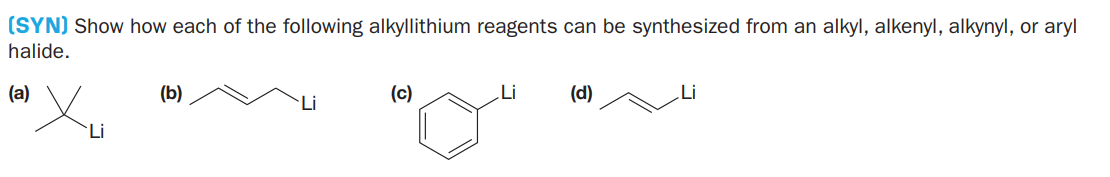
(SYN) Show how each of the following alkyllithium reagents can be synthesized from an alkyl, alkenyl, alkynyl, or aryl halide. (a) (b) (c) Li (d) Li Li `Li
Q: Which of the following is/are not feasible for the following ether synthesis?
A: Answers are explained Below
Q: Consider the structure of compound. Provide the a. most stable free-radical intermediate b. major…
A: Free Radical stability refers to the energy level of free radical .If the internal energy of free…
Q: Give a clear handwritten answer...Give a synthesis mechanism for given below reaction...
A: We have to synthesis the given compound. We also have to give the mechanism of it..
Q: Explain why A cross couples with the aryl iodide, rather than two molecules of A coupling together…
A: Aryl coupling is commonly a cross-coupling reaction. That means the reaction starts when two…
Q: If possible please answer all questions. Provide a complete, detailed curved-arrow mechanism for the…
A: Applying concept of Aldol Condensation reaction .
Q: The structure of the product, C, of the following sequence of reactions would be
A: The reaction of terminal alkyne with NaNH2 in ammonia causes the abstraction of the alkyne proton to…
Q: (SYN) Each of the following compounds can be produced (in low yield) from an alkyne, using a single…
A: Alkynes can undergo hydration reactions to form ketones (that tautomerises as enol as well) The…
Q: alkyl halide is most likely to A undergo the which forms a carbocation intermediate. reaction…
A: There are basically two types of substitution reactions ;- SN1 (nuclephilic substitution…
Q: Provide a Curved Arrow Mechanism for the following reaction. .CI HO- HCI
A: The above reaction is an example of a substitution reaction where the reaction goes through a…
Q: Give a clear explanation handwritten answer with textual explanation...give the mechanism of given…
A: The given reactant is a tertiary alkyl bromide which is an alkyl halide. The reagent is KCN and the…
Q: HO. orion °° Y
A: To find: mechanism of the given reactions.
Q: (b) ethanol, HCI соон heat
A: Esterification reaction: Carboxylic acid reacts with alcohol in the presence of an acid catalyst to…
Q: complete the reaction, show detailed mechanism
A:
Q: Provide the mechanism and product
A:
Q: Which of the following is most likely to be the first step in the general mechanism for…
A:
Q: An α carbon of a ketone or aldehyde can be alkylated or halogenated under basic conditions. Recall…
A: In the alpha halogenation reaction, an enolate formed from an aldehyde or ketone reacts with a…
Q: (a) Delineate the fate of reactions with the support of detailed the following mechanism:
A: i)This reaction proceeds with a mechanism like a benzyne as shown below. (As specified by…
Q: Please be certain to use good curved arrow notation for your mechanism. HYH2O HO,
A:
Q: Provide a curved arrow mechanism for the following reaction. NaCl CI
A: Nucleophilic substitution reaction is an organic reaction in which a nucleophile replaces another…
Q: Predict the major organic product of each of the following transformations. of кон
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Predict the most stable product structure by showing the detailed mechanism. Also write the reaction…
A: The above reaction is undergoing a thorough SN2 mechanism. Steric hindrance decreases the…
Q: Using the Malonester synthesis, write the synthesis of the following compound by showing its…
A: malonester synthesis is the chemical process in which the diethyl malonester is alkylated at the…
Q: Complete the following reactions, clearly indicating regio-and stereochemistry where applicable. In…
A: Aromatic oxidation of the side chain leads to the formation of carboxylic acids in the presence of…
Q: (a) For which aromatic compound do you expect nitration to take place faster: furan or thiophene?…
A:
Q: For the reaction shown, select the expected major organic product.
A: Electrophilic addition reactions : Addition of HX to alkene give alkyl halide is the product, it…
Q: Provide a reasonable mechanism using curved arrow notation for the following transformation, which…
A:
Q: Provide the full CURVED ARROW MECHANISM for the following reaction=
A: Types of intermediates in organic reactions : 1. Carbocation 2. Carbanion 3. Free radical…
Q: draw all mechanisms and resonance structures for this given organic structure
A: To solve this problem we have to write the mechanism of this reaction .
Q: Predict the organic and inorganic products of the given reaction, and select the class to which the…
A: It is an example of condensation reaction Here We are required to find the product of the reaction…
Q: Draw the curly arrow mechanism for the following reaction
A: The curly arrow mechanism for the following reaction is
Q: Complete the mechanism for the given Stork enamine reaction by drawing curved arrows, atoms, bonds,…
A: Carbonyl compounds react with a secondary amine to form an enamine. Enamines are nitrogen analogous…
Q: Give a clear explanation handwritten answer...give the mechanism of given bleow reaction
A: see below
Q: Provide a complete arrow push mechanism for each of the following reaction:
A: We have given the organic reaction we have to find the complete arrow push mechanism for each of…
Q: Predict the Mechanism which will inform stereo and regioselectivity on table given. Answer on…
A: The following mechanism and the bases and electrophile will be mentioned in a list given below. Also…
Q: ome me
A: 1. Step 1 : Birch reduction. Step 2 : The starting material is treated with acid results…
Q: True or false: Th application of the organometallic is depend on the metal type, if the M is main…
A: Given Th application of the organometallic is depend on the metal type, if the M ismain group use as…
Q: Predict the main product of each of the following reactions with their
A: a. alpha hydrogen containing aldehydes or ketone in presence of base to give aldol first, further…
Q: Given the compound shown, draw the acid chloride and nucleophile that will synthesize this compound.
A: The acid chloride and nucleophile that will synthesize the given compound is given below :
Q: Predict the major product of the following reactions and please provide reasoning throughout.
A: DBU (1,8-Diazabicyclo(5.4. 0)undec-7-ene) is a strong base. SN2 reaction will results the product…
Q: CH₂OH H
A: Here we have explain major product formed in the following given reaction.
Q: provide a synthesis of the target compound shown from the starting material that is provided,…
A: The reduction of carboxylic acid can be done by using a reducing agent such as Lithium Aluminium…
Q: Deuterium (D) is an isotope of H. Both D and H have one proton and one electron; H has no neutrons…
A:
Q: The major product/s that form/s during the nitration of benzenesulfonic acid is? choose the correct…
A: Please find your solution below : The nitration of benzene sulphonic acid is an example of…
Q: In cach case below select the synthetic procedure/s that could be used to carry out the…
A: Alkene iBH3iiH2O2,OH- antimarkovnikov's addition of H2O (via non-classical carbocation intermediate)…
Q: (SYN) Suggest how you would synthesize each of the following from phenylethanone (acetophenone),…
A:
Q: Provide a Curved Arrow Mechanism for the following reaction. .CI HO. HCI
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- Please answer question below, be very deatiled and draw out the mechanism reaction with arrowsprovide the detailed arrow pushing mechanism for the given reactions. Do not skip any stepPlease provide a detailed mechanism for the following transformation. Also, show the appropriate curved arrows to rationalize the next step. The quality of the electron pushing counts.
- . Use to curved arrow notation, propose a mechanism for the following reaction and state whether it is either SN1, E1, SN2, or E2. Give the IUPAC names of all organic reactants and products.Complete the curved arrow notation and include the missing organic intermediates for each of the four steps in the following nucleophilic aromatic substitution.Mechanism, in the space provided below please give the detailed mechanism for the following transformation
- Please provide a complete, detailed curved-arrow mechanism for the following reaction.Include ALL lone pairs and formal charges. Using the mechanism and a few words,explain the why the NaH deprotonates at the selected a-position and not the other a-position. Also explain why the indicated alkene is formed in the 3rd step rather than the other possible alkene product.(SYN) Draw the diene that would react with ethene to produce each of the following compoundsOrganic Chemistry Problem. Please help. Please provide neatly written mechanism using arrows to show electron flow and all intermediates. Thank you.

