Q: Show all the possible resonance structures. 5. a) b) N BO: 0
A: The movement of pi-electrons between atoms of a conjugated system is called delocalization. The…
Q: 6. 2 H₂ +0₂ → 2 H₂O 2(g) 2(g) For the reaction represented above at 25°C, what are the signs of AH°,…
A:
Q: of isomers? (a) CH3CH=CHCH₂CH3 (b) CH3CH₂OCH₂CH3 (c) (d) O CH3COCH₂CH3 and CH₂ /\ H₂C-CHCH₂CH3 and…
A: Given Different molecules
Q: 2Fe²+ + O₂+ 2 H+H₂O₂ + 2Fe³+ For the above redox reaction, assign oxidation numbers and use them to…
A: We have to identify the element which one is oxidised and which one is reduced. The given reactions…
Q: Provide the conjugate acids for the following bases: H3C-C NH₂: CH3CH₂OH
A: The conjugate acid of any substance will form after accepting the proton. Examples: H2O +…
Q: O MATERIAL/COMPOUND TESTED Copper(II) Oxide Potassium Nitrate Cobalt(II) Chloride Strontium Chloride…
A: As per the rules, only the first question (table) can be answered. For answers to the second table,…
Q: The activity coefficient (y) of a neutral molecule is typically approximated as 1.00; however, a…
A: The activity coefficient (γ) can be calculated using the following relation- log (γ) =kμ μ…
Q: 1. (a) Write balanced molecular equations for the following potential precipitation reactions.…
A:
Q: Calculate the reduced mass for the two isotopomers (HCl and HCI). Which isotopomer do you expect to…
A:
Q: Calculate the standard molar entropy of vaporization of water at 57.0 °C, given that its standard…
A: Given , Vaporisation of water .
Q: Build the following molecule with your model set and take a photo of a side view of your model in…
A:
Q: 4. Elizabeth is baking a loaf of bread and uses 48 grams of baking soda (NaHCO₂). How many molecules…
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: Write the number of valence electrons for each atom, total number of valence electrons, the Lewis…
A:
Q: [1] CH3CH₂ CH₂CH₂CH3 [2] CH3CH₂ CHCH₂CH3 TCH₂
A:
Q: Calculate the conc. of HCl in molarity having a 15.02 mL of HCl sol needed in a 26.1 mL of 0.32…
A:
Q: A buffer solution is made by dissolving 0.45 moles of a weak acid (HA) and 0.33 moles of KOH into…
A:
Q: What is the relationship between the structures shown? Selected answer will be automatically saved.…
A: First structure has molecular formula : C8H18 Second structure has molecular formula: C8H16 Thus…
Q: b. (6.0241 X 1.24) / (.0147 X 10.0) = c. The density of an object that has a mass of 16.0 grams and…
A: Density is the ratio of mass of the object to its volume.
Q: CI Ill F LL F F F H 2. H
A:
Q: Provided that Palm Spring contains 20 mg/liter of Cu2+, given that it has no precipitation in water,…
A: Given Amount of Cu2+ = 20 mg/L Amount of Cu2+ left = 0.5 mg/L Amount of Cu2+ reacted = 20-0.5 =…
Q: E. A 20.0 mL aliquot of a 0.515 M aqueous solution is added to 125 mL of pure water. What is the…
A: In this problem, we are asked to find the concentration of a new solution after adding a 20.0 mL…
Q: An unknown acid named HXO, is discovered and determined to be a weak acid. Which of the following is…
A: Acidic Salt forms when strong acid neutralizes with weak base. Basic salt forms when strong base…
Q: A chemist carefully measures the amount of heat needed to raise the temperature of a 0.27 kg sample…
A: Amount of heat,Q required to raise the temperature of a substance having no. of moles ,n and molar…
Q: A student dissolves 14.2 g of ammonium chloride (NH4C1) in 200. g of water in a well-insulated open…
A:
Q: Give the IUPAC name of this compound
A: We have to determine the IUPAC name of the given compound
Q: QUESTION 11 A molecular compound has the formula C11H1601 How many grams of this compound would you…
A: Given- Molecular formula-C11H16O1 Molar mass =164 gm
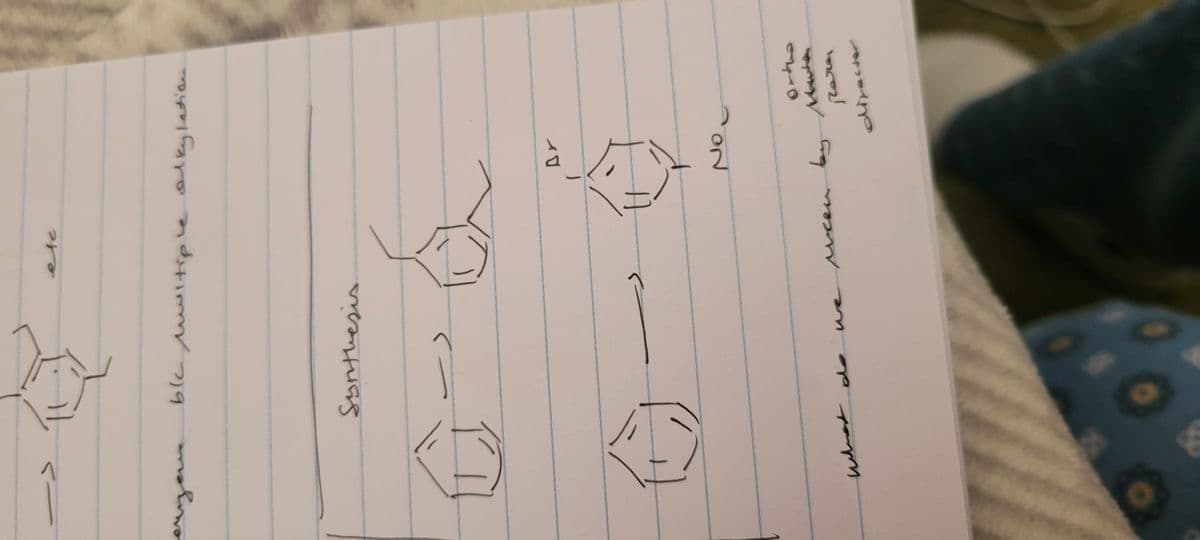
Q: 1. The following are just individual steps from different mechanisms. Fill in the curved arrows for…
A: curly arrows are drawn on the structural formulae of reactants in a chemical equation to show the…
Q: Which of the following can inhibit nitrification? 1. Low HNO2 2. High NH3 3. High NH4+ 4. pH < 5…
A: The nitrification process is the microbial oxidation of ammonia (NH3) to nitrite (NO2-) and then to…
Q: Rank the following compounds in order of decreasing acidity, putting the most acidic first. I on A)…
A: Need to rank the compound in order of decreasing acidity. Need to determine the structure of…
Q: A 100.0-mL sample of brackish water was made ammoniacal, and the sulfide it contained was titrated…
A: The stoichiometry of the reaction between silver nitrate and sulfide ions is: 2Ag++S2-→Ag2S(s) This…
Q: Q4. A city's water supply is fluoridated by adding NaF. The desired concentration of F is 1.6 ppm.…
A: To calculate the amount of NaF needed to achieve the desired concentration of F- in the water, we…
Q: What is the total formal charge for the molecule shown below? Hint: Start by identifying individual…
A: Formal Charge (qF) = Nv-Nnb-Nb2 Where, Nv , Nnb , Nb are the number of valance electrons, nonbonding…
Q: A mixture of three noble gases (He, Ne and Ar) is confined to a 3.24 L container at 24 °C. The He…
A:
Q: Draw the structure(s) of the product(s) expected if 1-methylcyclohexanol were treated with…
A: GIVEN 1-methylcyclohexanol
Q: 45. Which is the most reactive carbonyl compound? A) I B) II C) III D) IV ļ W
A:
Q: Bioactive form of the drug thalidomide Identify the absolute configuration of the stereocenter in…
A: Cahn Ingold Prelog rules : 1) Give the priority to the substituents attached at chirality center…
Q: Rank the following structures in increasing order of their boiling points flowest one on top…
A: Boiling point is related to the bonding and surface area of contact between the two molecule, on…
Q: 2. Propose an arrow-pushing mechanism and product for the following series of reactions. (First,…
A: 2. Given that, a reaction scheme is shown below We have to give the product with the arrow pushing…
Q: Name this molecule by taking into account the stereochemistry.
A: Introduction Compounds are formed when two or more elements are chemically combined. Nomenclature is…
Q: 9. How many hydrogen atoms are there in 500.0 L of water? (dH₂O = 1.00 g mL-¹) A: 3.34 x 1028
A: The question asked for the number of hydrogen atoms in 500.0 L of water, given a density of 1.00…
Q: ality al, g. on te ew led ed in X= A solution was prepared by dissolving 140.0 g of KCl in 315 g of…
A:
Q: Draw the skeletal structure of 3- methylheptane from the condensed formula (shown below).…
A: Given: condensed formula
Q: The heat of vaporization AH, of heptane (C₂H₁6) is 31.2 kJ/mol. Calculate the change in entropy AS…
A: Explanation: The chemical equation follows: C7H16 (l) ⇌ C7H16(g) • To calculate the number…
Q: How manipulation of protonation state of compounds influence solubility in organic and aqueous…
A: The solubility of a compound in different solvents is an important property that can be influenced…
Q: Solid aluminum (Al) and oxygen (O₂) gas react to form solid aluminum oxide (Al,0₁). Suppose you have…
A:
Q: 103 +51 +6H+ + 3 12 + 3 H₂O 1₂ +1 → 13 C6H8O6 +13+ C6H6O6 + 2 H+ + 31
A: A chemical equation is said to be balanced if the moles of same type of atoms are equal on either…
Q: . What is the product of the following reaction? wana A. Heat B. میکرونی علمی کوٹے D. 35
A:
Q: Suppose you are performing a gas-producing reaction to experimentally determine the molar volume of…
A: Given Vapor pressure of water= 19.8mmHg Atmospheric pressure= 768.6 Temperature= 22°C = 273+22 =…
Q: Aqueous hydrochloric acid (HCI) will react with solid sodium hydroxide (NaOH) to produce aqueous…
A:
Q: Supposed that the concentration of H+ in Solution #1 is 10,000 times greater than in Solution #2.…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Plastic photochromic sunglasses are based on the following reversible rearrangement of a dye inside the lenses that occurs when the lenses are exposed to sunlight. The original dye absorbs UV light but not visible light and is thus colorless, while the rearrangement product absorbs visible light and is thus darkened. (a) Show the mechanism of the rearrangement. (b) Why does the rearrangement product absorb at a longer wavelength (visible light) than the original dye (UV)?(need answer in step by step) Deaw major product of followinv reavtion, make sure to draw enantiomers if expected1. When you add H-Br in a terninal alkene, the product has the rule od Markovnikov? 2. The type pf intermetary thag forms when we add HgSO4/H2SO4/H2O in an alkyl is? Please solve my both questions.thank you.



