Q: arning A current of 3.77 A is passed through a Pb(NO3)₂ solution for 1.70 h. How much lead is plated…
A:
Q: IZ OH -OH H3O (trace) H2O ? IZ HO HN. -OH но- HN. -OH
A:
Q: Determine the molecular formulas given the following empirical formulas and molar masses: bd KS0…
A: GivenEmpirical Formula = KSO4Molar mass = 270.32 g/molMolecular Formula = To be determined
Q: In the 1.0 M CUSO4 solution, the following species are present: Cu²+, SO42, H₂O, H3O+, and OH. Based…
A: Set up 1 observation: When CuSO4 gets dissociated, then the ions would be Cu+2, SO4-2 and water…
Q: What is the mass of 4.0x10" formula units of potassium chloride (KCI)?
A:
Q: draw a Bohr-Rutherford diagram to show electron transfer during reaction of beryllium metal and…
A: Ionic bond - A bond formed by transfer of electrons between a metal and non metal called ionic bond.…
Q: Which of the following species are capable of hydrogen bonding among themselves? Select all that…
A: we have to select the species which can hydrogen bond with itself
Q: Which of the following is an example of a covalent bond? A) K-CI B) Al - CI C) H - CI D) AI-AI
A: Given bonds: K-Cl Al-Cl H-Cl Al-Al
Q: At what temperature (in °C) does the root mean square speed of CO2 molecules equal 565.0 m/s?…
A:
Q: 3. Give the mechanism (using curved-arrow notation) to account for the following reaction. CH₂OH H*…
A: Tertiary carbocation is more stable than secondary carbocation and secondary carbocation is more…
Q: Metal block 1 and block 2 at temperatures TK and 27 K, respectively, were allowed to equilibrate to…
A: This is a problem of second law of thermodynamics which gives definition of entropy. Here , two…
Q: Calculate the number of oxygen molecules required to produce 16.1 g KHCO3. Express your answer in…
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: Calculate the pH in a 4.8x10-3 M solution of HI
A:
Q: Determine the percentage composition
A:
Q: 89 mol of an ideal gas has a pressure of 2.53 atm and a volume of 68.59 L, what is the temperature…
A:
Q: A chemist fills a reaction vessel with 0.969 atm nitrogen (N₂) gas, 6.95 atm oxygen (O₂) gas, and…
A:
Q: 17.4 Solubility and..... Gibb E 18.3 Gibbs Free E... 5.3 Enthalpies of.... ENTROPY AND FREE ENERGY…
A:
Q: What is the IUPAC name for the following compound? CH₂=CH-CH=CH-CH=CH-CH₂-CH3 Submit Answer Retry…
A: Organic compound are compound which have different functional group present and they are named using…
Q: Determine the volume of hexane that contains 5.33 × 1022 molecules of hexane. The density of hexane…
A: Please go through my hand written notes for the solution. The answer is 11.6 mL of hexane.
Q: A voltaic cell consists of a Pb/Pb2+Pb/Pb2+ half-cell and a Cu/Cu2+Cu/Cu2+ half-cell at 25 °C°C. The…
A:
Q: IR spectrometer is often purged with drv nitrogen
A: Nitrogen is an inert gas. It will not cause any effect on the spectrometer when it is not working or…
Q: Rank the following aqueous solutions in order of decreasing freezing point. Select 1 (highest…
A: Colligative properties: The colligative properties are those properties of solutions that depend on…
Q: If 0.42 mg calcium fluoride, CaF2 is used as above to add fuoride to the drinking water, does CaFz…
A: Given that 0.42 mg CaF2 is used to add fluoride in drinking water.We have to find between 0.42 mg of…
Q: 5. How many grams per milliliter of NaCI are contained in a 0.250 M solution? FW = 58.4 mg/mmol)
A: Molarity is defined as number of mole of solute divide by volume of solution in liter
Q: Chemists can produce silver metal by reacting copper metal with a solution of silver nitrate: Cu(s)…
A: Given -> Cu(s) + 2AgNO3(aq) -----> 2Ag(s) + Cu(NO3)2(aq) Mole of Copper= 0.24 mole Mole of…
Q: A balloon is inflated to a volume of 7.0 L on a day when the atmospheric pressure is 765 mmHg. The…
A: According to Boyle's Law, at constant temperature, volume of any fixed amount of gas is inversely…
Q: . Determine the best answer on the follow: What buffer pH can be prepared from an arbitrary weak…
A: 2. Buffer of weak acid is composed of weak acid and its salt of strong base. Buffer system is…
Q: Calculate the enthalpy change, A, H, for the formation of 1.00 mol of magnesium carbonate (also used…
A: Here, we have to calculate the enthalpy change for the formation of 1.00 mol of magnesium carbonate…
Q: Chemistry Draw the organic products obtained in each reaction. Hna IJNICE: 1221 131H₂O, A OE! [1]…
A: "Since you have asked multiple-subparts questions, we will solve the first three questions for you.…
Q: Part A Calculate the standard enthalpy change for the reaction where the heats of formation are…
A: Standard enthalpy of reaction is given by: ∆Hrxn0 = ∆Hf0(Products) - ∆Hf0 (reactants) ∆Grxn0 =…
Q: Part 1. Mechanisms. Consider the following reactions. y CH3CH₂OH H₂SO4 OCH₂CH3 OH Redraw the…
A: This is an example of epoxide ring opening in acidic medium. In acidic medium the ring opening…
Q: What is the IUPAC name of the following compound? CH3 снажено OH
A: IUPAC name consist of three parts that is prefix , word root and suffix. Prefix represents…
Q: Calculate the volume of 12M HCl (density = 1.185 g/mL containing 36.31% by mass of HCl) necessary to…
A: The Volume of HCl necessary to react with the amount of borax can be calculated using the following…
Q: Assuming equal concentrations, arrange these solutions by pH. Highest pH Ca(OH)₂ (aq) NaOH(aq)…
A: Given compounds: Ca(OH)2(aq) NaOH(aq) HClO4(aq) NH3(aq) HBrO(aq) We have to arrange these solutions…
Q: Which kinds of additive should be used to decrease the rigidity of PS? a. A lubricant b. A…
A: Here the key point is to check the low molecular weight polymer. The polymer which is having low…
Q: A chemist fills a reaction vessel with 7.76 atm nitrogen (N₂) gas, 7.51 atm hydrogen (H₂) gas, and…
A:
Q: Draw the major product for each of the Draw hydrogen atoms on chirality centers. Reaction A Reaction…
A: Alcohol after reaction with HBr gives carbocation which undergoes rearrangement to produce more…
Q: Draw the cycloalkene that would react with the reagent given to account for the product formed. O +…
A:
Q: Predict which of the following liquids has greater surface tension: acetone (CH3COCH3) or…
A: The attraction forces between the different molecules are known as intermolecular forces. In general…
Q: Calculate the total change in entropy, when a sample of nitrogen gas of mass 28 g at 298 K and 1.00…
A: This problem is based on Thermodynamics. Here we have to find out the entropy of the systems via…
Q: 1. A solution of a weak acid was tested with the following indicators and the colors noted: Methyl…
A: Indicators are the substance added to the titration mixture to determine the endpoint accurately as…
Q: The chemical equation for the complete combustion of propane, C₂Hg(g), is C₂Hg(g) +5 0₂(g) → 3…
A: The chemical equation given is C3H8 (g) + 5 O2 (g) → 3 CO2 (g) + 4 H2O (g) Mass of propane, C3H8 (g)…
Q: One mole of a perfect gas expanded isothermally and reversibly at 0 °C from 1 to 1/10 bar. Calculate…
A:
Q: A typical car battery contains 8.16 kg of lead. Determine the number of lead atoms in this battery.…
A:
Q: 7. Determine the cell potential (in V to two decimal places) for the following electrochemica cell:…
A: Given that an electrochemical cell is Cu(s)|Cu2+(aq, 0.02 M)||Cu2+(aq, 0.50 M)|Cu(s). We have to…
Q: The equilibrium constant for the formation of Cu(CN)42- is 2.0 x 1030. Calculate the value of pCu2+,…
A: we have to calculate the value of pCu2+
Q: The solubility product, Ks, of Cd3(PO4)2 is 2.5 x 10-33. What is the solubility (in g/L) of…
A: we have to calculate the solubility (in g/L) of Cd3(PO4)2 in pure water
Q: Part 2. Predict the Organic Products. Predict the major organic product(s) for each of the following…
A:
Q: Calculate the heat of reaction AH for the following reaction: CH₂(g) + 2O₂(g) →CO₂(g) + 2 H₂O(g) You…
A:
Q: What quantity in moles of hydrogen gas at 150.0 °C and 23.3 atm would occupy a vessel of 8.50 L?
A: Given Pressure( P) = 23.3 atm Volume (V) = 8.50 L Temperature (T) = 150.0 oC Number of moles (n) = ?
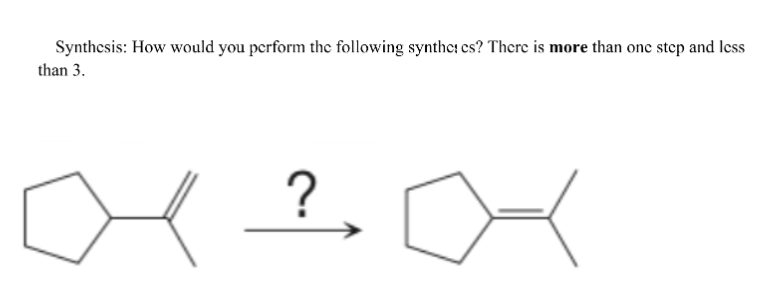
Step by step
Solved in 2 steps with 2 images

- this is a multiple step synthesis. you must provide all necessary reaction conditions (HBr, HgSO4, etc.) and the products produced after each step of the synthesis.Help! Need the reagents and mechanism for the following synthesis. Also, how did it become meta directing? Thanks!Draw the neutral organic starting material. The Hint: The reaction conditions support electrophilic addition of Br2 to an alkene's C=C double bond, which would normally yield a dibromo product. However, the product has only one bromine atom, with a C–O bond on the adjacent carbon. This fragment is diagnostic for a halohydrin, where an oxygen nucleophile (water or alcohol) reacts with a bromonium intermediate to generate the O–CH2CH2–Br motif. Work backwards to determine what the starting material must look like. There should be an alcohol and an alkene in the neutral organic starting material.
- hi, I need help to explain what is going on all those steps in this synthesis and also u nee to k ow the names of the produkt.USE INFORMATION ONLY LEARNED IN ORGANIC CHEM 1 Please illustrate the 2 substrates and reagent necessary to synthesize the following products:Please help with the following ochem reaction... show the mechanism for the following reaction and what the major product(s) would be

