t affect the rate of a le temperature, ors (orientation), and ch changes with the factors that ummarized in an s equation: /RT A is the frequency mergy, the gas constant, rature. - 1 of 1 A certain reaction has an activation energy of 67.0 kJ mol-¹ and a frequency factor of A₁ = 3.80×10¹2 L mol-¹s 1. What is the rate constant, k, of this reaction at 20.0 °C ? S Express your answer with the appropriate units. Indicate the multiplication of units explicitly either with multiplication dot (asterisk) or a dash. k = Value Submit Request Answer Units The linear form of the Arrhenius equation The Arrhenius equation can be rearranged to a form that resembles the equation for a straight line: Ink = -(/) (+) + In A R E BAK ? Y m x + b ,a is, and b is the y-intercept or In A. The linearity of this equation is where y is In k, m is the slope or illustrated graphically in the image. (Figure 1)
t affect the rate of a le temperature, ors (orientation), and ch changes with the factors that ummarized in an s equation: /RT A is the frequency mergy, the gas constant, rature. - 1 of 1 A certain reaction has an activation energy of 67.0 kJ mol-¹ and a frequency factor of A₁ = 3.80×10¹2 L mol-¹s 1. What is the rate constant, k, of this reaction at 20.0 °C ? S Express your answer with the appropriate units. Indicate the multiplication of units explicitly either with multiplication dot (asterisk) or a dash. k = Value Submit Request Answer Units The linear form of the Arrhenius equation The Arrhenius equation can be rearranged to a form that resembles the equation for a straight line: Ink = -(/) (+) + In A R E BAK ? Y m x + b ,a is, and b is the y-intercept or In A. The linearity of this equation is where y is In k, m is the slope or illustrated graphically in the image. (Figure 1)
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter11: Chemical Kinetics: Rates Of Reactions
Section: Chapter Questions
Problem 25QRT
Related questions
Question
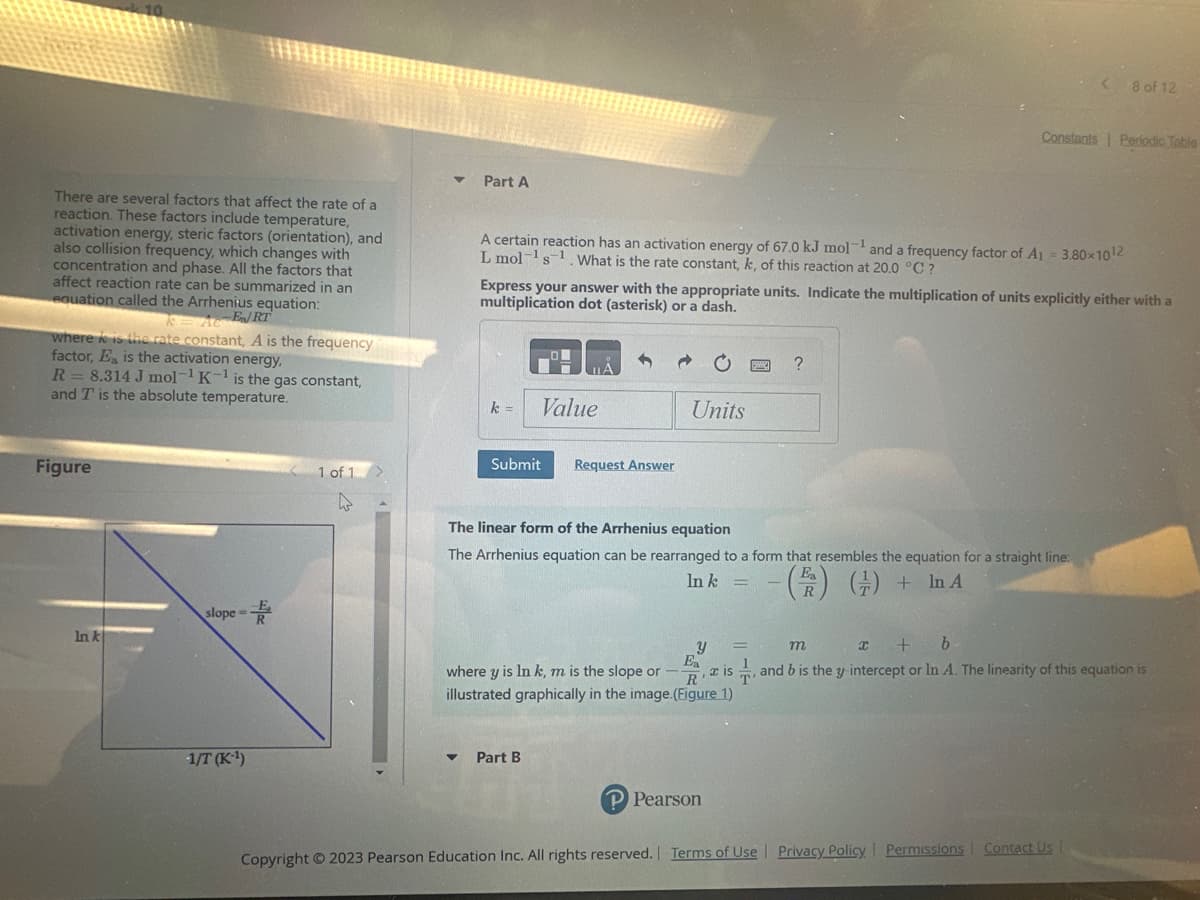
Transcribed Image Text:There are several factors that affect the rate of a
reaction. These factors include temperature,
activation energy, steric factors (orientation), and
also collision frequency, which changes with
concentration and phase. All the factors that
affect reaction rate can be summarized in an
equation called the Arrhenius equation:
KA-E/RT
where is the rate constant, A is the frequency
factor, Ea is the activation energy,
R = 8.314 J mol-¹ K-¹ is the gas constant,
and T is the absolute temperature.
Figure
In k
slope=
-1/T (K-¹)
1 of 1
▼
Part A
▼
A certain reaction has an activation energy of 67.0 kJ mol-¹ and a frequency factor of A₁ = 3.80×10¹2
L mol-¹ s 1. What is the rate constant, k, of this reaction at 20.0 °C?
k =
Express your answer with the appropriate units. Indicate the multiplication of units explicitly either with a
multiplication dot (asterisk) or a dash.
Value
Submit
Request Answer
Part B
Units
Y
a is
where y is In k, m is the slope or
R'
illustrated graphically in the image. (Figure 1)
The linear form of the Arrhenius equation
The Arrhenius equation can be rearranged to a form that resembles the equation for a straight line:
In k
() + In A
E
P Pearson
?
<
Constants Periodic Table
=
8 of 12
m
Т + b
1
and b is the y-intercept or In A. The linearity of this equation is
Copyright © 2023 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Policy Permissions | Contact Us
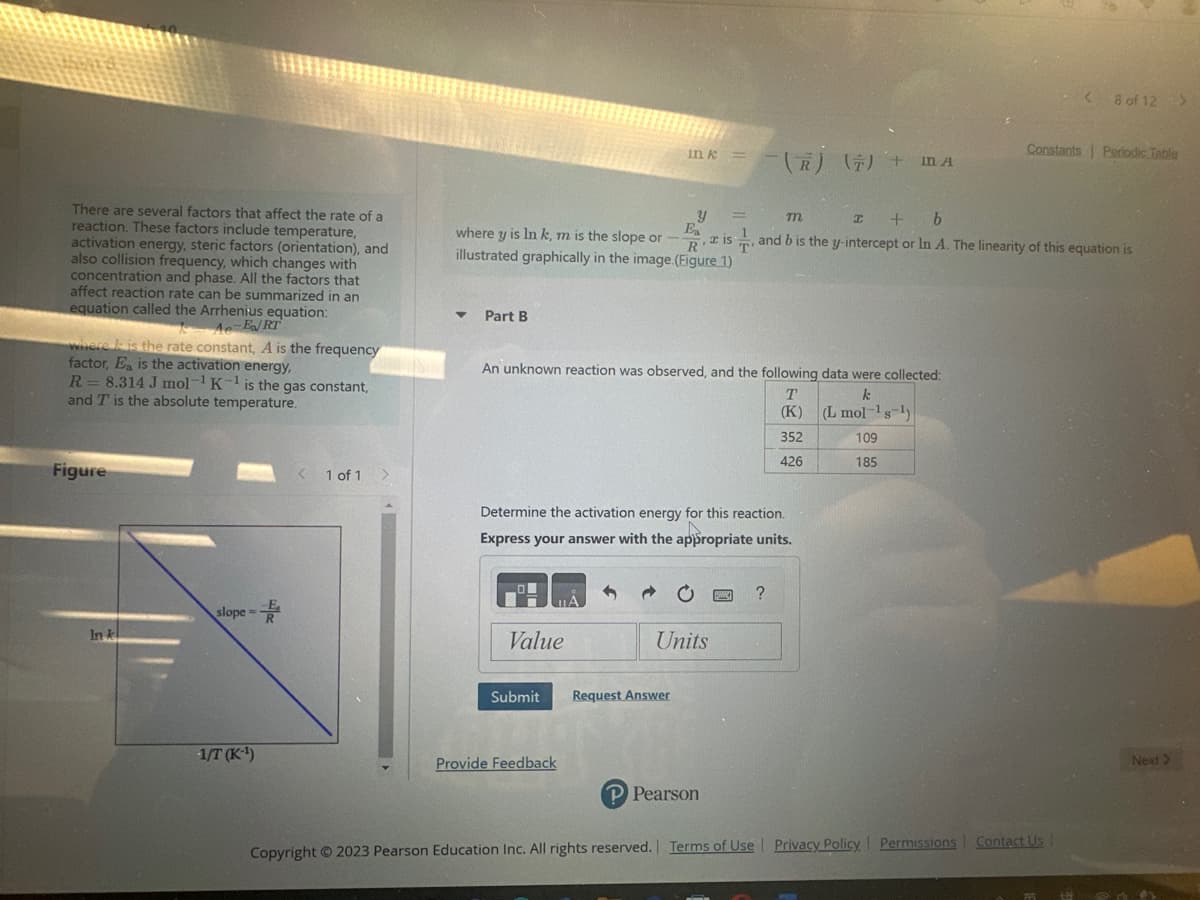
Transcribed Image Text:There are several factors that affect the rate of a
reaction. These factors include temperature,
activation energy, steric factors (orientation), and
also collision frequency, which changes with
concentration and phase. All the factors that
affect reaction rate can be summarized in an
equation called the Arrhenius equation:
Ae-E/RT
where is the rate constant, A is the frequency
factor, Ea is the activation energy,
R = 8.314 J mol-¹ K-¹ is the gas constant,
and T is the absolute temperature.
Figure
In k
slope=
-1/T (K-¹)
1 of 1
▼ Part B
Ea
where y is In k, m is the slope or -- , a is
illustrated graphically in the image. (Figure 1)
Value
Submit
An unknown reaction was observed, and the following data were collected:
k
(L mol-¹s ¹)
109
185
Provide Feedback
A
in k
Determine the activation energy for this reaction.
Express your answer with the appropriate units.
1
Y
Ĉ
Request Answer
Units
(R) (7) + MA
P Pearson
m
I + b
and b is the y-intercept or In A. The linearity of this equation is
?
T
(K)
352
426
Copyright © 2023 Pearson Education Inc. All rights reserved. Terms of Use | Privacy Policy Permissions
< 8 of 12 >
Constants Periodic Table
Contact Us
Next >
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning