Biomedical Instrumentation Systems
1st Edition
ISBN:9781133478294
Author:Chatterjee
Publisher:Chatterjee
Chapter9: Instrumentation In Blood Circulation
Section: Chapter Questions
Problem 13P
Related questions
Question
Hi I need help creating a graph of my my data. Thank you so much!

Transcribed Image Text:b. Place a "0" in the middle of the y axis. Choose
appropriate scales.
c. Label the axes of the graph: Determine dependent and
independent variables, and place each on the appropriate
аxis.
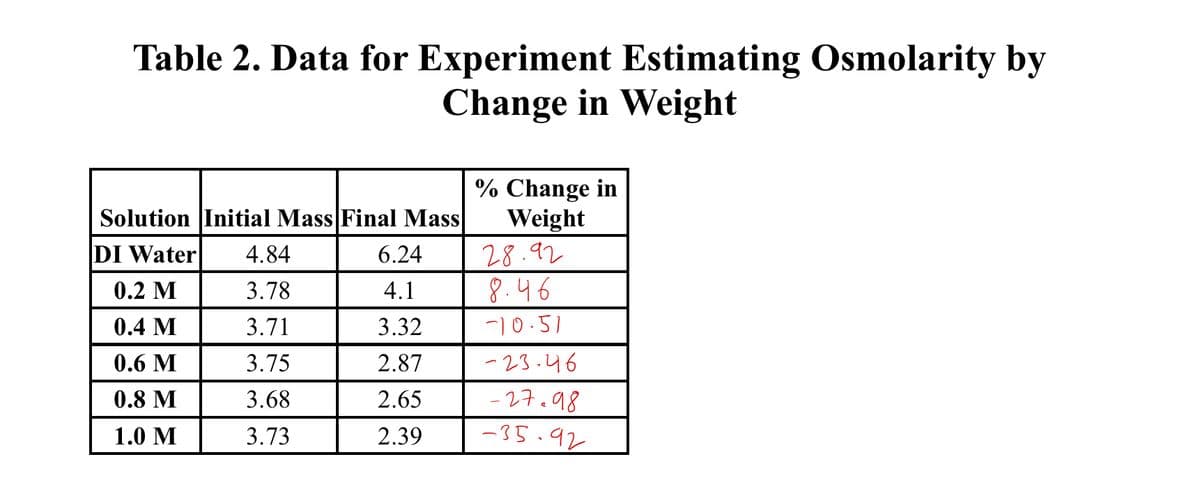
Transcribed Image Text:Table 2. Data for Experiment Estimating Osmolarity by
Change in Weight
% Change in
Weight
28.92
8.46
Solution Initial Mass Final Mass
DI Water
4.84
6.24
0.2 M
3.78
4.1
0.4 M
3.71
3.32
-)0.51
0.6 M
3.75
2.87
-23.46
0.8 M
3.68
2.65
27.98
1.0 M
3.73
2.39
-35.92
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you



Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning



Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning