Table 5G.1 Solubility constants at 298.15 K Compound Formula K, Aluminium hydroxide Al(OH), 1.0 x 10-33 Antimony sulfide Sb,S, 1.7 x 10-93 Magnesium ammonium phosphate Barium carbonate BaCO, 8.1 x 10-9 MGNH,PO. 2.5 x 10-13 fluoride BaF, 1.7 x 10-6 M9CO, MgF, carbonate 1.0 x 10-5 sulfate BasO, 1.1 x 10-10 6.4 x 10-9 fluoride Bismuth sulfide Bi,S, CaCO, 1.0 x 10-97 hydroxide Mg(OH)2 1.1 x 10-11 Calcium carbonate 8.7 x 10-9 Mercury(l) chloride Hg,Cl, 1.3 x 10-18 fluoride CaF2 4.0 x 10-11 Hg,l2 HgS black: iodide 1.2 x 10-28 hydroxide Ca(OH)2 CaSO, 5.5 x 10-6 Mercury(ll) sulfide 1.6 x 10-62 sulfate 2.4 x 10-5 red: 1.4 x 10-53 Copper(I) bromide CuBr 4.2 x 10-8 Nickel(II) hydroxide Ni(OH), 6.5 x 10-18 chloride CuCl 1.0 x 10-6 Silver bromide 7.7 x 10-13 AgBr Ag,CO, AgCI A9OH iodide Cul 5.1 x 10-12 carbonate 6.2 x 10-12 sulfide Cu,S 2.0 x 10-47 chloride 1.6 x 10-10 Copper(II) iodate Cu(1O,)2 CuC,04 1.4 x 10-7 hydroxide 1.5 x 10-8 oxalate 2.9 x 10-8 iodide Agl 1.5 x 10-16 sulfide Cus 8.5 x 10 45 sulfide Ag,S Zn(OH), 6.3 x 10-51 Iron(II) hydroxide Fe(OH)2 1.6 x 10-14 Zinc hydroxide 2.0 x 10-17 sulfide Fes 6.3 x 10-18 sulfide ZnS 1.6 x 10-24 Iron(III) hydroxide Fe(OH), PbBr, 2.0 x 10-39 Lead(II) bromide 7.9 x 10-5 chloride PbCl, 1.6 x 10-5 PBF2 Pb(IO3)2 Pbl2 fluoride 3.7 x 10-8 iodate 2.6 x 10-13 iodide 1.4 x 10-8 sulfate 1.6 x 10-8 sulfide PbS 3.4 x 10-28
Table 5G.1 Solubility constants at 298.15 K Compound Formula K, Aluminium hydroxide Al(OH), 1.0 x 10-33 Antimony sulfide Sb,S, 1.7 x 10-93 Magnesium ammonium phosphate Barium carbonate BaCO, 8.1 x 10-9 MGNH,PO. 2.5 x 10-13 fluoride BaF, 1.7 x 10-6 M9CO, MgF, carbonate 1.0 x 10-5 sulfate BasO, 1.1 x 10-10 6.4 x 10-9 fluoride Bismuth sulfide Bi,S, CaCO, 1.0 x 10-97 hydroxide Mg(OH)2 1.1 x 10-11 Calcium carbonate 8.7 x 10-9 Mercury(l) chloride Hg,Cl, 1.3 x 10-18 fluoride CaF2 4.0 x 10-11 Hg,l2 HgS black: iodide 1.2 x 10-28 hydroxide Ca(OH)2 CaSO, 5.5 x 10-6 Mercury(ll) sulfide 1.6 x 10-62 sulfate 2.4 x 10-5 red: 1.4 x 10-53 Copper(I) bromide CuBr 4.2 x 10-8 Nickel(II) hydroxide Ni(OH), 6.5 x 10-18 chloride CuCl 1.0 x 10-6 Silver bromide 7.7 x 10-13 AgBr Ag,CO, AgCI A9OH iodide Cul 5.1 x 10-12 carbonate 6.2 x 10-12 sulfide Cu,S 2.0 x 10-47 chloride 1.6 x 10-10 Copper(II) iodate Cu(1O,)2 CuC,04 1.4 x 10-7 hydroxide 1.5 x 10-8 oxalate 2.9 x 10-8 iodide Agl 1.5 x 10-16 sulfide Cus 8.5 x 10 45 sulfide Ag,S Zn(OH), 6.3 x 10-51 Iron(II) hydroxide Fe(OH)2 1.6 x 10-14 Zinc hydroxide 2.0 x 10-17 sulfide Fes 6.3 x 10-18 sulfide ZnS 1.6 x 10-24 Iron(III) hydroxide Fe(OH), PbBr, 2.0 x 10-39 Lead(II) bromide 7.9 x 10-5 chloride PbCl, 1.6 x 10-5 PBF2 Pb(IO3)2 Pbl2 fluoride 3.7 x 10-8 iodate 2.6 x 10-13 iodide 1.4 x 10-8 sulfate 1.6 x 10-8 sulfide PbS 3.4 x 10-28
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter12: Chemical Equilibrium
Section12.3: Determining Equilibrium Constants
Problem 12.3PSP
Related questions
Question
Use the data given to estimate the molar solubilities of (a) BaSO4, (b) Ag2CO3, (c) Fe(OH)3, (d) Hg2CI2 in water.
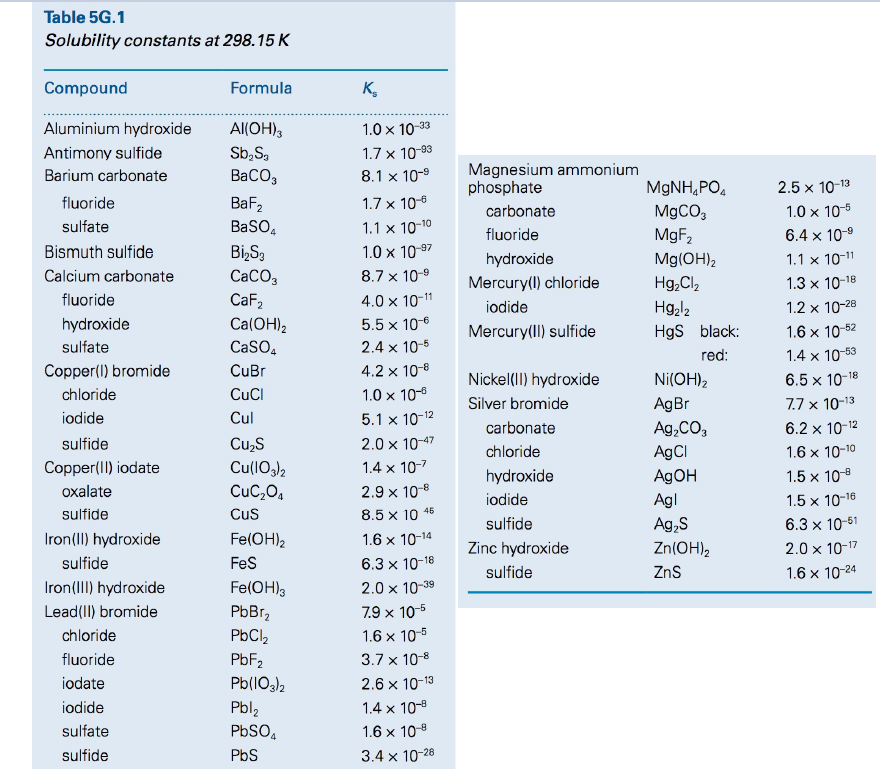
Transcribed Image Text:Table 5G.1
Solubility constants at 298.15 K
Compound
Formula
K,
Aluminium hydroxide
Al(OH),
1.0 x 10-33
Antimony sulfide
Sb,S,
1.7 x 10-93
Magnesium ammonium
phosphate
Barium carbonate
BaCO,
8.1 x 10-9
MGNH,PO.
2.5 x 10-13
fluoride
BaF,
1.7 x 10-6
M9CO,
MgF,
carbonate
1.0 x 10-5
sulfate
BasO,
1.1 x 10-10
6.4 x 10-9
fluoride
Bismuth sulfide
Bi,S,
CaCO,
1.0 x 10-97
hydroxide
Mg(OH)2
1.1 x 10-11
Calcium carbonate
8.7 x 10-9
Mercury(l) chloride
Hg,Cl,
1.3 x 10-18
fluoride
CaF2
4.0 x 10-11
Hg,l2
HgS black:
iodide
1.2 x 10-28
hydroxide
Ca(OH)2
CaSO,
5.5 x 10-6
Mercury(ll) sulfide
1.6 x 10-62
sulfate
2.4 x 10-5
red:
1.4 x 10-53
Copper(I) bromide
CuBr
4.2 x 10-8
Nickel(II) hydroxide
Ni(OH),
6.5 x 10-18
chloride
CuCl
1.0 x 10-6
Silver bromide
7.7 x 10-13
AgBr
Ag,CO,
AgCI
A9OH
iodide
Cul
5.1 x 10-12
carbonate
6.2 x 10-12
sulfide
Cu,S
2.0 x 10-47
chloride
1.6 x 10-10
Copper(II) iodate
Cu(1O,)2
CuC,04
1.4 x 10-7
hydroxide
1.5 x 10-8
oxalate
2.9 x 10-8
iodide
Agl
1.5 x 10-16
sulfide
Cus
8.5 x 10 45
sulfide
Ag,S
Zn(OH),
6.3 x 10-51
Iron(II) hydroxide
Fe(OH)2
1.6 x 10-14
Zinc hydroxide
2.0 x 10-17
sulfide
Fes
6.3 x 10-18
sulfide
ZnS
1.6 x 10-24
Iron(III) hydroxide
Fe(OH),
PbBr,
2.0 x 10-39
Lead(II) bromide
7.9 x 10-5
chloride
PbCl,
1.6 x 10-5
PBF2
Pb(IO3)2
Pbl2
fluoride
3.7 x 10-8
iodate
2.6 x 10-13
iodide
1.4 x 10-8
sulfate
1.6 x 10-8
sulfide
PbS
3.4 x 10-28
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 6 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning