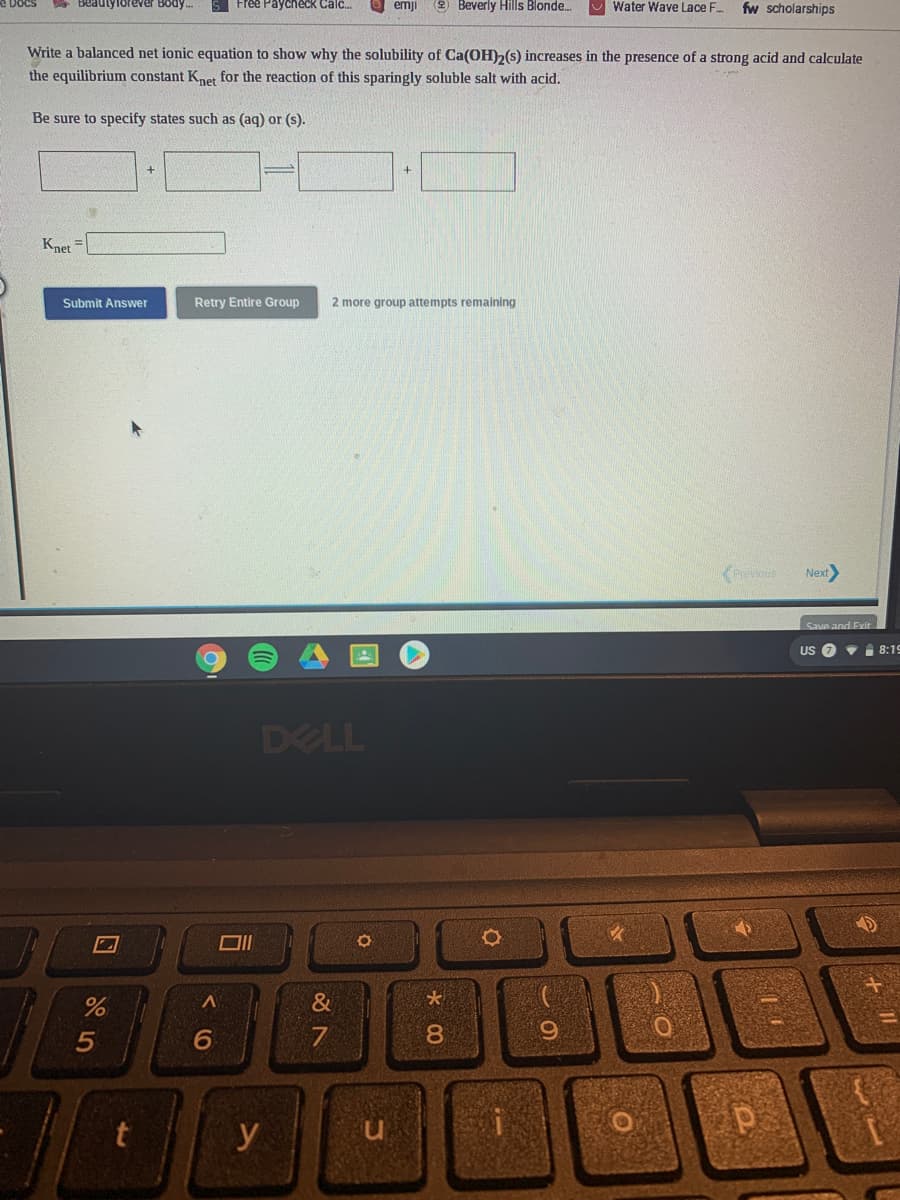
Write a balanced net ionic equation to show why the solubility of Ca(OH)2(s) increases in the presence of a strong acid and calculate the equilibrium constant Knet for the reaction of this sparingly soluble salt with acid. Be sure to specify states such as (aq) or (s). Knet
Write a balanced net ionic equation to show why the solubility of Ca(OH)2(s) increases in the presence of a strong acid and calculate the equilibrium constant Knet for the reaction of this sparingly soluble salt with acid. Be sure to specify states such as (aq) or (s). Knet
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 12.60PAE
Related questions
Question
15a.
Knet needs to be a number
Transcribed Image Text:Beaulylorever Body.
Free Paycheck Calc.
emji
2 Beverly Hills Blonde.
Water Wave Lace F..
fw scholarships
Write a balanced net ionic equation to show why the solubility of Ca(OH)2(s) increases in the presence of a strong acid and calculate
the equilibrium constant Knet for the reaction of this sparingly soluble salt with acid.
Be sure to specify states such as (aq) or (s).
Knet =
Submit Answer
Retry Entire Group
2 more group attempts remaining
Prevous
Next
Save and Exit
US 7
i 8:19
DELL
&
6.
7
8.
9.
y
n.
口
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning