Q: There are two benzylic bromides that give (Z)-3,5-dimethyl-2-phenyl-2-hexene on E2 elimination. Draw…
A:
Q: These reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O,…
A: 1) BH3, THF, H2O2 - It is hydroboration oxidation reaction in which terminal alkyne reacted with BH3…
Q: II. Name or write the structures of the following: a. (E,E) 3,5-0octadiene b. c. H;C=CHCH=CHC=CH OH…
A:
Q: true or false. There is nothing wrong with the reaction below
A:
Q: 3. Chemical Formulas and Thermodynamics (continued) C. From the bond energy data given below, (a)…
A: #3: #(a): The chemical formula of formic acid is HCOOH or CH2O2. The standard state elements, C, H,…
Q: Compound Z absorbs light of wavelength 320 nm. A 1.0 x 10-3 mol dm-3 solution of a compound Z gives…
A:
Q: 45.0 g of lead (Il) acetate is reacted with 63.0g of ammonium sulfide, how many particles of product…
A:
Q: a. Calculate the mmoles of l created on the reaction? 103 + 3 + H2O Reaction: b. The triiodide…
A:
Q: (E,E) 3,5-0ctadiene
A: Detail description is given below
Q: what is the term symbol for the ground state of O2-
A: Given that, ion = O2- Term symbol for the ground state = ?
Q: What can be done to lower the activation energy? Question 6 options: increasing the…
A: "Catalysts " can lower the activation energy and increase the reaction rate without being consumed…
Q: Reaction rate can NOT be increased by Question 17 options: adding an effective catalyst to the…
A:
Q: (s) NAOH EŁOH H, COOEE dietil malonato
A:
Q: Cl2 uv or heat
A: In presence of sunlight at alpha position cholorination take place
Q: reactant and calculate the theoretical yield of water producet ced in the experiment, what is the…
A:
Q: 1.Two samples of the same soil is subjected to pH analysis using a pH meter, the first is analysed…
A: While measuring the pH of a soil, water or KCl can be used as the diluent.
Q: what is the hybridization of each carbon atom in toluene and how many S orbitals are present within…
A:
Q: DQUESTION 1, THREE (3) moles of H2(2) and THREE (3) moles of I2(2) are introduced into a 2L…
A:
Q: Sketch the titration curve when 10.0 mL of a 0.20 M solution of the polyprotic weak acid H2CO3 is…
A: H2CO3 is a weak diprotic acid. When it is titrated with NaOH, we see two inflection points.
Q: The phenolphthalein indicator used in this fatty acid test is pink in basic solution and colorless…
A: ✓As we know,phenolphthalein is an indicator that is pink in alkaline solutions of about pH10. ✓When…
Q: Find the pH of a 0.0025 M HCl solution.
A: pH of a solution = -log[H+]
Q: QUESTION 22 1. To separate NH3 (MW = 17.0304) and mn3. (AM = 54.9380) in mass spectrum, what is the…
A: A mass spectrometer is an instrument that is used to measure the mass of particles. The resolution…
Q: Sodium hydroxide, NaOH is not a Bronsted-Lowry base but its aqueous solution is basic. Provide a…
A:
Q: OH :CH2 H3C H,C + A, В > H
A:
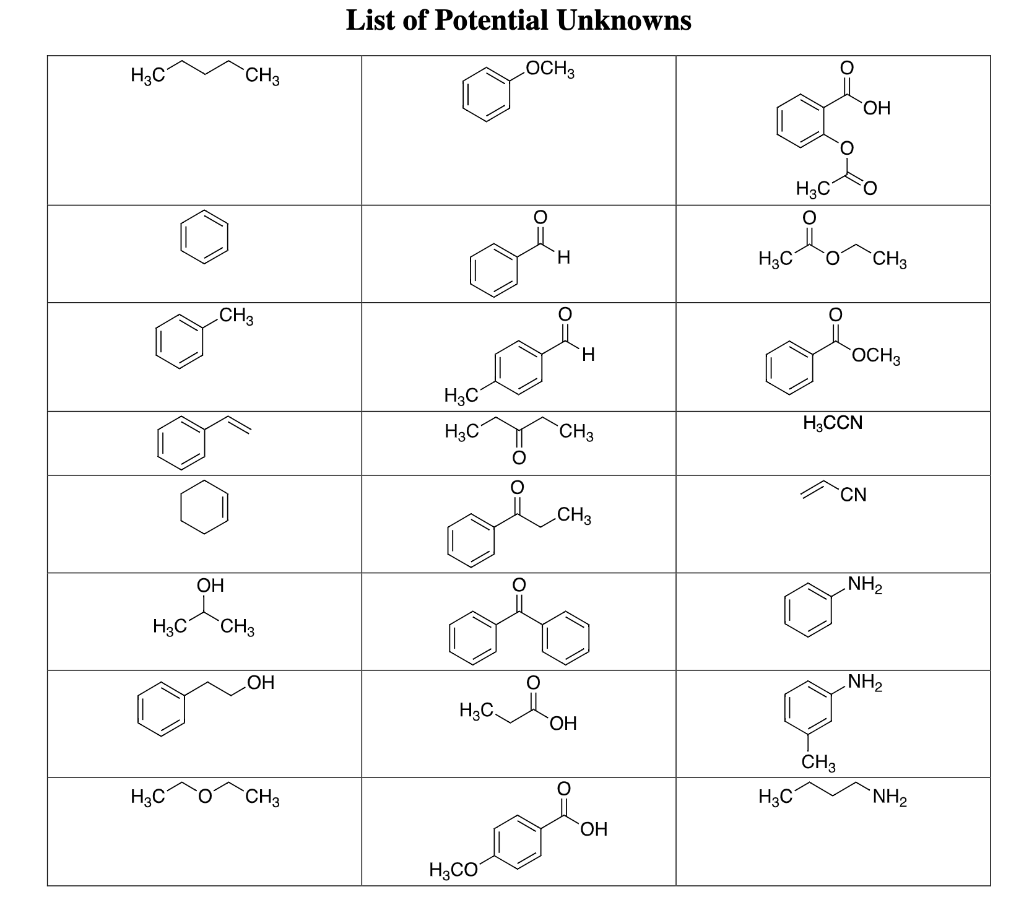
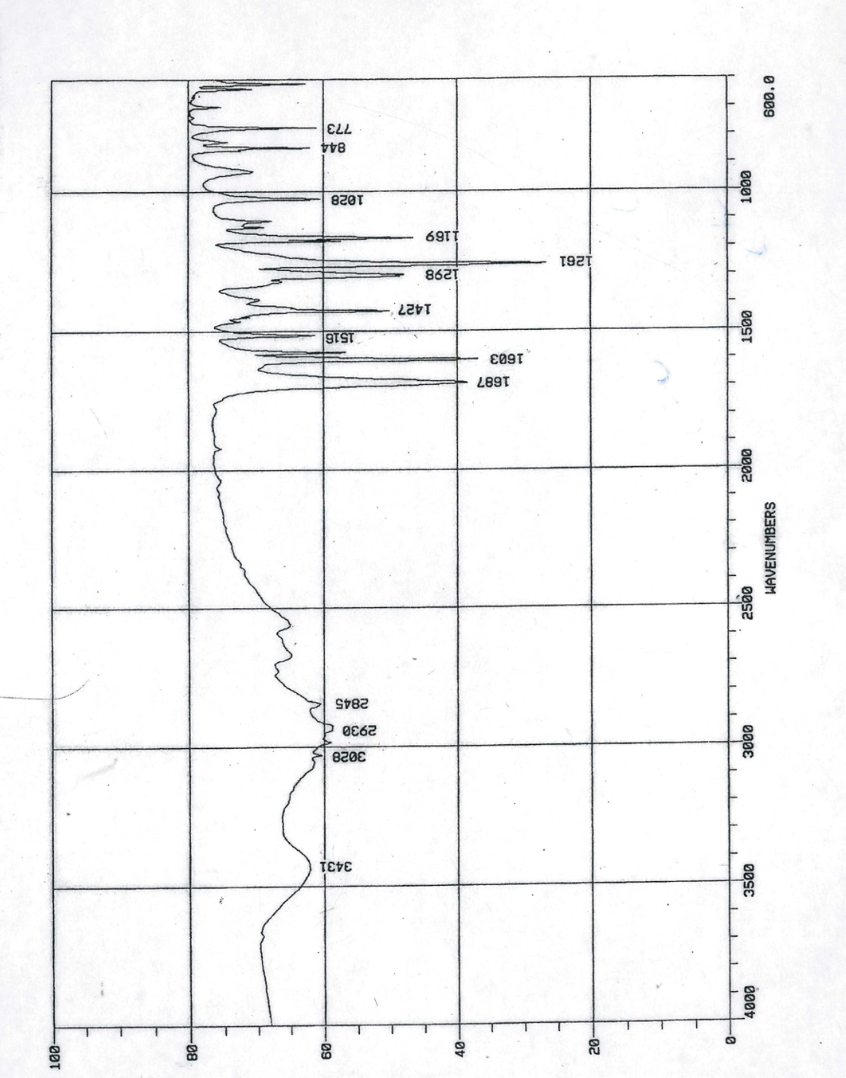
Q: In each of the items below, determine/choose which group does it belong. Aldehyde or Ketone 1.…
A: Aldehyde and ketone both are organic compounds.Functional group present in aldehydes is -CHO and…
Q: A 0.400 L sample of gas at - 55.0 degrees * C and a pressure of 2.15 atm is allowed to expand to…
A:
Q: Chemistry 1) write data on this spectrum 275nm 85anm Peaentage ↑ of absoabance
A: The given data is uv absorption spectra.It gives two maxima at 275nm and 350nm.
Q: 88. Copper and bromine react according to this equation: Cu (s) + Br, () → CuBr, (s) If 0.39 moles…
A: Cu(s) + Br2(l) ....> CuBr2(s) Given that: Moles of Cu = 0.39 moles Moles of Br2 = 0.25 moles
Q: 4. Calculate the AH for the following reaction: NO 0 > NO2 Given the following information: 2 03 > 3…
A: NO +O ---> NO2
Q: Draw the curved arrow that explains how the products are formed. Select Draw Rings More Erase ++…
A:
Q: Question 33 DNA can best be described as A. two backbones of alternating deoxyribose and nitrogenous…
A: 32. When the soaps disperse in water they form spherical clusters known as micelles these micelles…
Q: Which of the following reagent best accomplish this transformation below? Which of the following…
A:
Q: Calculate the pressure of one mole of an ideal gas which occupies 12.0 L at 25°C.
A: We know the relation ; PV = nRT ( P is pressure,V is volume, n is mole number, R is gas constant,T…
Q: 2) When a certain freon used in refrigeration was expanded adiabatically from an initial pressure of…
A: The explanation nedd hand written,not possible in type format,as some symbol not available. I think…
Q: A Fajans titration of a 0.7908-g sample required 45.32 mL of 0.1046 M AgNO3 . Express the results of…
A:
Q: Filling the Blanks. Fill in the blanks with the correct word/words to complete the given statement.…
A: 1) Carbohydrates provide energy to the body, particularly through glucose, a simple sugar that is a…
Q: How much heat is put into 100g of water to raise temperature from 25.0 to 75.0°C? The specific heat…
A:
Q: -CH3 1. Hg(OАc)2, Н,о 2. NABH,
A:
Q: Explain why casein is not denatured by the addition of acid (unlike most proteins).
A: Denaturation of proteins means the breaking of bonds (hydrogen bonds) within the protein molecule…
Q: d. 65°C = K 187 K = C
A:
Q: * OWLV2 |Online teaching and le. keCovalentActivity.do?locator=assignment-take a 白☆*ロ( Update…
A: Fe2+(aq) + H2(g) ----> Fe(s) + 2H+(aq)
Q: Study the following reaction scheme and give a detailed reaction mechanism that leads to the…
A:
Q: CH-CE CH CH2-C-CH3
A:
Q: the 4th row, what is written above the the arrow? cl2 and
A:
Q: छोल्सद पह CHormie सपलtm तिम्यणा पप उ०. P৮N6 + ctmmp०৪ Pb3০५+ CU2०3+Mmd.tNo
A:
Q: 96. Methane gas (16 g, 1.0 mole) and oxygen gas (96 g, 3.0 moles) are reacted in a sealed container.…
A:
Q: What are the units of the rate constant for a first ordereaction? M-sec M sec sec O M2
A: Which one of the following is correct answer
Q: I. Identify the compounds (reactants or products) corresponding to each letter. If there are several…
A: The given reactions are basically electrophilic addition to alkynes. Much like alkenes, alkynes…
Q: 5) Discuss the physical interpretation of any one Maxwell relation.
A: Maxwell equation describes How a electromagnetic field behaves in a generel medium. The main…
Q: Analyze the given benzene ring structure and choose the correct IUPAC name of the structure.…
A: From the given structure we see that at 3 position ethyl group,at 2 position methyl and at 1…
Step by step
Solved in 2 steps with 1 images

- Please show steps and answer accordingly. Thanks! X3A.Aleks data for AgBrO3 is 5.38 x 10^-51. Which of the following compounds can form hydrogen bonds with water?a. octaneb. HClc. NaCld. acetic acid2. Maria wants to determine the percent water content of an unknown hydrate of AB. Given the following data:mass of empty crucible - 12. 2784gmass of AB · xH20 + crucible - 17.4960gmass of AB + crucible - 15.1314gCalculate the % water content of the hydrate3. From the above data, give the correct formula for the hydrate. (AB₂ Molar mass = 129.84 g/mol)
- just give me the finally anwer thank youuuQUESIONS 33 AND 34 PLEASERecall that the formula for ΔHrxn = [(-Ccal)(moles of limiting reactant)/(ΔT)]If the actual concentration of the sodium hydroxide used in the calibration part of the experiment was lower than the stated value, will the value of Ccal increase, decrease, or remain the same? Please explain. How about if the actual concentration of the sodium hydroxide used in the calibration part of the experiment was lower than the stated value. However, the NaOH solution used in the determination of ΔHrxn was of the correct concentration. Will the magnitue of ΔHrxn increase, decrease, or remain the same? Please explain.
- Which of the following is a spontaneous reaction.? a. Rxn with ΔH =- 10Kj/mol ΔS= -5J/mol T= 300K b. NaCl +H20 -> NaOH + HCl 25C c. H20(l) -> H2O(s) Temp: 25C d. Dissolution of 100g of solid sugar in 100 mL ice tea. Consider following reaction: HgO (s) -> Hg(l) + ½ O2 (g) Delta H = +90.7 kj/mol. What quantity of heat in kj/mol is required to produce one mole HgO? Write your answer without units. Given the following data 2ClF(g) + O2(g) --> Cl2O(g) + F2O (g) Delta H= 167.4 kJ I 2ClF3(g) + 2O2(g) --> Cl2O(g) + 3F2 O (g) Delta H= 341.4 kJ II 2 F2(g) + O2(g) ---> 2F2O (g) Delta H= -43.4 kJ III Calculate the delta H in kJ for below reaction: ClF(g) + F2(g) ---> ClF3(g)Calculate the ΔG°rxn using the following information. 2 H2S(g) + 3 O2(g) → 2 SO2(g) + 2 H2O(g) ΔG°rxn = ? ΔH°f (kJ/mol) -20.6 0.00 -296.8 -241.8 S°(J/mol∙K) 205.8 205.2 248.2 188.8The standard enthalpies of formation of ClO and ClO2 are101 and 102 kJ/mol, respectively., Calculate the overallenthalpy change for each step in the following catalyticcycle: What is the enthalpy change for the overall reaction thatresults from these two steps? ClO(g) + O3(g) ---->ClO2(g) + O2(g) ClO2(g) + O(g) ---->ClO(g) + O2(g)

