Q: Na20 CO SiC SO2
A: An ionic compound is formed when a cation and ion combine via electrostatic force of…
Q: A nephrolith is formed due to Ca (II) oxalate, phosphate or urate precipitation. Will CaC204…
A:
Q: Mass of Inner cup with stirrer on the digital balance=36.7 Mass of Inner cup with stirrer and warm…
A:
Q: calculate iodine value . Fatty acid Molecular Formula % 16:0 C16H32O2 13.2 16:1 C16H3002 0.2 18:0…
A: The iodine value of an oil is a measure of the degree of unsaturation which is defined as the number…
Q: Calculate the pH of a 0.200 M solution of NH,CI. The K, value of ammonia is 1.8x10-S and…
A: Given, [NH4Cl] = 0.200 M Ionization constant of NH3 = Kb = 1.8 x 10-5 Calculate the pH = ?
Q: The triphenylmethyl radical is highly delocalized by resonance. In the resonance hybrid, how many…
A: There are ten different positions have radical character
Q: Name this compound according to the system we went over in lecture. CH3 CH3
A:
Q: Calculate the equilibrium constant for the overall reaction at this temperature. 2) Calculate…
A:
Q: Using 2-methylcyclopentanol and any appropriate reagents, show stepwise reaction how the following…
A: In the first question subpart c), we will show the stepwise reaction by using proper reagent and in…
Q: all possibilities drawn out please
A:
Q: प्रोचक नेी कर वष्मामामरवमवेद্ উেoddे छ एिer के अ्रष केए उन्.। C EXPlaim A (6) ACHAळP
A: The short string of amino acids are commonly known as peptides. They are comprising 2–50 amino…
Q: Mass of Inner cup with stirrer on the digital balance=36.7 Mass of Inner cup with stirrer and warm…
A:
Q: 1. Why is acetanilide no longer available as a drug in the market? 2. Why is the synthesis of…
A:
Q: PPh + R. +R. PPh RBr oc- Br Ph,P PhP čo explain how's the hybridization? prove your answer
A: To solve this question, it is quite important to determine the oxidation state and the proper…
Q: What is the de Broglie wavelength of Usain Bolt with a mass of 79 kg and a speed of 11 m/s? nm What…
A:
Q: Calculate E°cell a
A:
Q: Loving to another question will save this response. 19 complex [Co(NH3)6l absorbs light at a…
A: Given,the complex [Co(NH3)6]2+ absorbs light at a wavelength 465nm. Calculate the ligand field…
Q: At a certain temperature this reaction follows first-order kinetics with a rate constant of 0.657…
A:
Q: 3. Which aqueous solution is expected to have the highest vapor pressure? Assume that all of them…
A:
Q: A precipitate dissolves if: Select one: а. Q=Ksp b. Q= 2*Ksp C. Q>Ksp- d. Q<Ksp
A: Ksp is the solubility product of salt . Q is the ionic product of salt. If ionic product is equal to…
Q: A chemist dissolves 643. mg of pure perchloric acid in enough water to make up 250. mL of solution.…
A: Given, mass of perchloric acid = 643 mg = 0.643 g…
Q: Selected Standard Molar Enthalpies of Formation ΔΗ; ΔΗ; (kJ/mol) | Substance | (kJ/mol) Substance…
A: Given ,that the reaction is- 4 NH3(g) + 5 O2(g) → 4 NO(g) + 6 H2O(g) Calculate the enthalpy for the…
Q: (b) Supported lipid bilayers (SLBS) are widely used in biophysical research to investigate the…
A: The answer is provided below-
Q: Would you increase or decrease the pressure (via a volume change) of each of the following reaction…
A:
Q: 2. A cell can be prepared from copper and tin. What is the Ecel for the cell that forms from the…
A:
Q: AMoving to another question will save this response. stion 6 For the following coordination complex…
A:
Q: Calculate both saponification and iodine value? Fatty acid Molecular Formula 16:0 C16H32O2 13.2 16:1…
A: The saponification value is the number of milligrams of potassium hydroxide required to neutralize…
Q: 8. The compound Mg(OH)2 is classified as an Arrhenius base because, when dissolved in water, there…
A: According to the Arrhenius theory, the substance that gives hydrogen ion H+ in the aqueous solution…
Q: The donor species in a complex is called a ligand (A) True B) False
A: A complex compound or coordination compound consists of a central metal with vacant orbitals which…
Q: A slab of lithium (Li) weighs 1.49 X 10^3mg and dimensions of 20.9mm, 11.1mm, and 11.9mm. A)…
A: Given that - Weight of lithium ,Li = 1.49×103 mg = 1.49 g = 0.00149 kg Dimension of slab = 20.9…
Q: The system is in equilibrium, if : Select one: а. the concentrations of reactants are equals 0.1…
A:
Q: Question 4 How much energy is rekased by the decay of I gram of 230th of I gramm of 230 in the…
A:
Q: The reaction scheme below shows the reactions of alkene B. Reaction II Reaction I Reaction III C + D…
A:
Q: A cell can be prepared from copper and tin. What is the E call for the cell that forms from the…
A:
Q: g)A chemical reaction has ΔH° = +250kJ/mol. Which of the following statements is/are true concerning…
A: ∆H° sign is positive that means Reaction is Endothermic because energy is absorbed by the system.…
Q: Question 16 For the compound Name the compound Identify each chiral conter Are goo metric posible?…
A: In order to write iupac nomenclature we have to follow some rules of iupac 1) first select…
Q: "When 0.774g sample of C7H 5N 30 6 is burned in a bomb calorimeter, the temperature increases from…
A: Given ,that the sample=0.774g of C7H5N3O6 is burned in a bomb calorimeter Temperature increases from…
Q: Write the reaction involved in Ferrox Test. a. What is the species responsible?…
A:
Q: In the lab, you determined the wavelength whose absorbance was maximum. That wavelength was used to…
A: A question based on tools in analytical chemistry that is to be accomplished.
Q: equation AH (kJ/mol) 2AB(s) + C2D(1) - 2CB(g) + A2D(s) 521 21 ED(g) + ED2(g) + A2D(s) 2AED2(s) -430…
A:
Q: Question 16 atient is given a O.05 mg dose of 99 Te, which has half tife of 6hours, How long will it…
A:
Q: The magnetic susceptibility, lefi, of a sample of solid K3COF, at room temperature as measured using…
A: The answer is attached below-
Q: 1. Og 23. 2. H202 A. B. C. 애
A: In this question we have to tell the product of the reaction.
Q: Using the data below, calculate the entropy for the formation of rust, iron(II) oxide from Fe and…
A: In this question we have to tell the entropy of the reaction.
Q: State the functional group that will be produced in the reaction below. heat CH3-CH2-CH2-COOH + NH3…
A:
Q: Question 14 SHOW WORK Given the following reaction: Cac 2(s) + 2 H 20-C2H 2(g) + Ca(OH) 2(aq) Assume…
A:
Q: What are reactants will form the enamine shown? For the following molecules classify them as…
A:
Q: Redox is a portmanteau of reduction and oxidation A True B False
A: Given statement, Redox is a portmanteau of reduction and oxidation.
Q: Under certain conditions the rate of this reaction is zero order in ammonia with a rate constant of…
A: Given, 2NH3(g) ➝ N2(g) + 3H2(g) Zero order kinetic. Rate constant (k) = 0.0097 Ms-1 Volume of the…
Q: How would you prepare the following from propan-1-ol? H3C .CI H3C H3C ОН
A: The given problem is based on functional group conversions in organic chemistry.
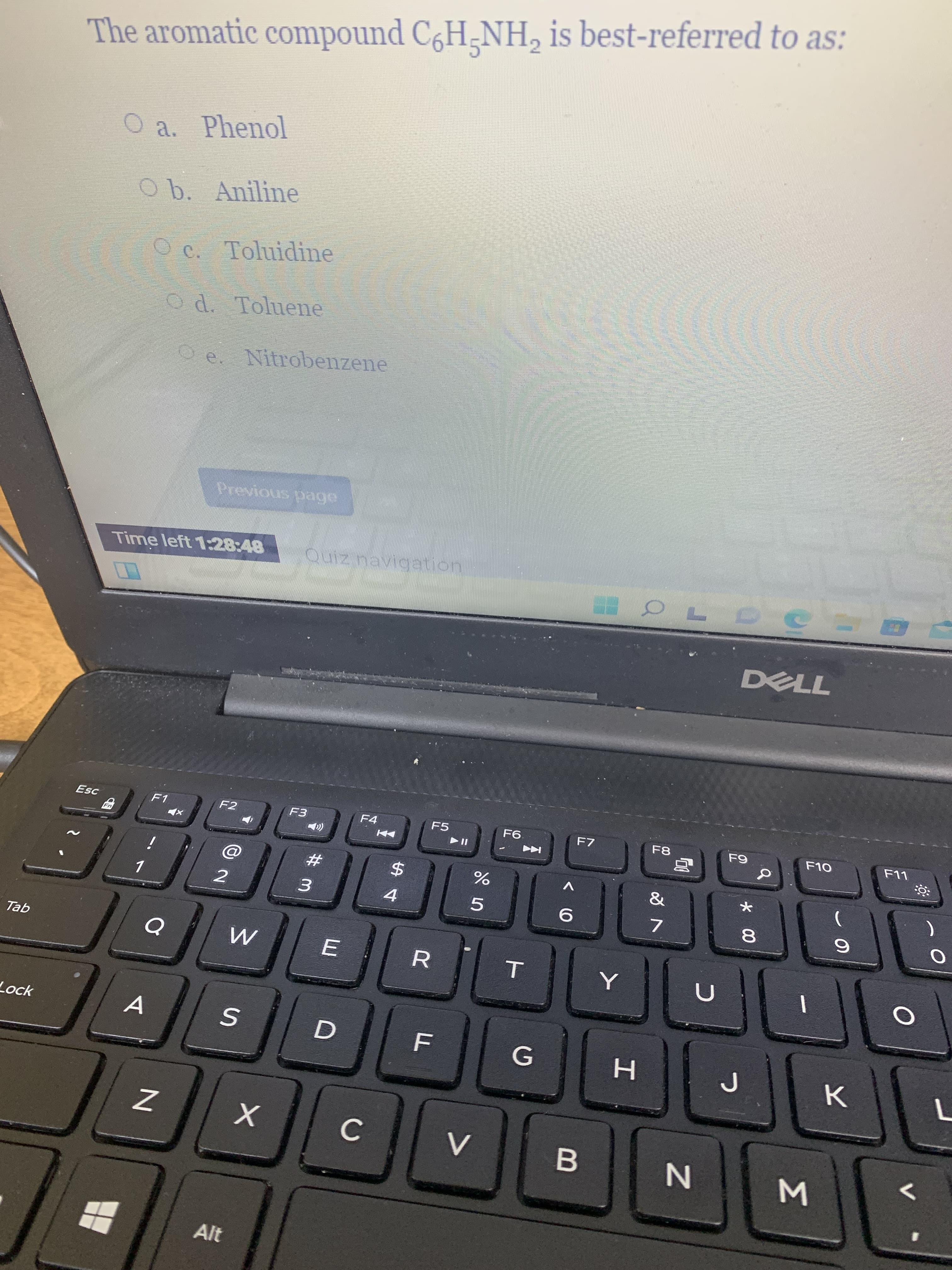
Step by step
Solved in 2 steps with 2 images

- 1 i. What is Resonance Theory? Sate five conclusions that can be drawn from the theory. ii. State the two main experiments that were used to establish the extra stability of the benzene molecule. iii. What are the factors that confer Aromaticity to an organic molecule? iv. State the effects of substituents on a benzene derivative towards further aromatic substitution. V. Based on the above suggest the various types of substituents that can be attached to Benzene.Is the following statemente true or false? "The pKa of a compound will be big if its acid form is aromatic."Choose one answer for the following. Refer to the diagram below. 1. Hybridization of atom labelled A (sp, sp2, or sp3) 2. Hybridization of atom labelled B (sp, sp2, or sp3) 3. Hybridization of atom labelled C (sp, sp2, sp3) 4.Class compound for functional group D (phenols, aryl halide, alcohols, aromatics, or carboxylic acids) 5. Class compound for functional group E (phenols, aryl halide, alcohols, aromatics, or carboxylic acids)
- ANSWER ALL QUESTIONS1 What is Resonance Theory? Sate five conclusions that can be drawn from the theory.2.State the two main experiments that were used to establish the extra stability of the benzene molecule.3.What are the factors that confer Aromaticity to an organic molecule?4.i. State the effects of substituents on a benzene derivative towards further aromatic substitution. ii.Based on the above suggest the various types of substituents that can be attached to Benzene.How do you write the nomenclature? Also, how do you do R and S?Wich of the following alternstives is responsible for the following structures to be non-aromatic? A) cyclic structure B)planarity C)hunckle rule D)conjugation E)none of the above
- Give good hand written explanation.Looking at the structure of naphthalene given below, determine if it is aromatic or not. Explainthe reasons for your answer?What steps would you need to take to calculate the pKa1 and pKa2 of something? For example: hydrosulfuric acid H2S(aq) (-38.6 ΔH^0 f kJ mol^-1).
- The calicene molecule has a different dipolar moment of 0 and also thedipole points to the larger ring. Draw a structure resonance where you can see that distribution of loads and using the concept of aromaticity and resonance structures, explain why the moment dipolarit's different from zeroWhat is the structure based on the HNMR?Determine the class of the compound, which contains only carbon and hydrogen, and exhibits the infrared spectrum below. Possible compound classes are:alkane, alkene, alkyne, aromatic, alcohol, amine, aldehydeketone, carboxylic acid, acid chloride, ester, amide Chem 1 = Chem 2 image=

