The attached image shows the different amounts and concentrations for an iodide clock experiment, showing the effects of persulfate and iodide concentrations on reaction rate. How do I fill up this table? Please provide a general formula/solution for solving for the Molarity portion.
The attached image shows the different amounts and concentrations for an iodide clock experiment, showing the effects of persulfate and iodide concentrations on reaction rate. How do I fill up this table? Please provide a general formula/solution for solving for the Molarity portion.
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter11: Chemical Kinetics: Rates Of Reactions
Section: Chapter Questions
Problem 36QRT
Related questions
Question
The attached image shows the different amounts and concentrations for an iodide clock experiment, showing the effects of persulfate and iodide concentrations on reaction rate.
How do I fill up this table? Please provide a general formula/solution for solving for the Molarity portion.
| Runs | [S2O8 2-], M | [I–], M | [S2O3 2-], M | Reaction time,s | Rate, M/s |
| 1 | 49.10 | ||||
| 2a | 106.10 | ||||
| 3 | 197.44 | ||||
| 4 | 69.20 | ||||
| 5 | 53.05 |
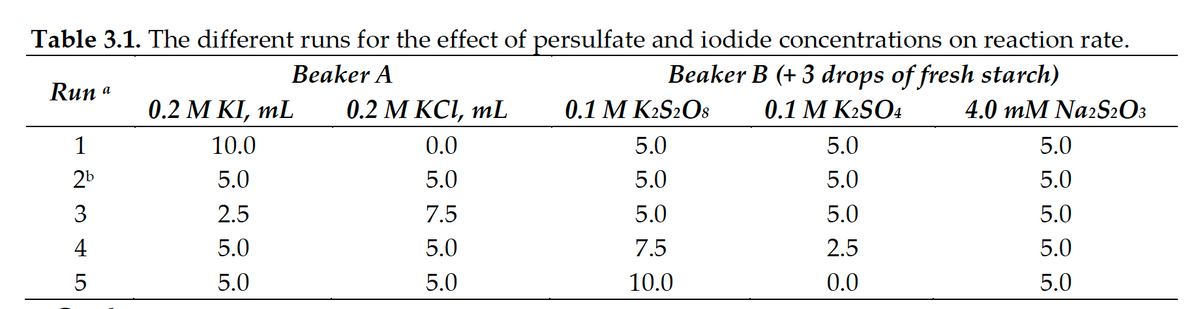
Transcribed Image Text:Table 3.1. The different runs for the effect of persulfate and iodide concentrations on reaction rate.
Beaker B (+ 3 drops of fresh starch)
Beaker A
Run a
0.2 Μ KΙ mL
0.2 М КСІ, mL
0.1 M K2S2O8
0.1 M K2SO4
4.0 mM Na2S2O3
1
10.0
0.0
5.0
5.0
5.0
2b
5.0
5.0
5.0
5.0
5.0
3
2.5
7.5
5.0
5.0
5.0
4
5.0
5.0
7.5
2.5
5.0
5
5.0
5.0
10.0
0.0
5.0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning