The circles in the diagrams below represent energy levels in an atom, and the arrows show electron transitions from one energy level to another. The spacing between circles is proportional to the differences in energy. Rank the atoms based on the energy of the photon that is absorbed, from lowest to highest. ONLY rank those transitions that absorb a photon and that are physically possible.
The circles in the diagrams below represent energy levels in an atom, and the arrows show electron transitions from one energy level to another. The spacing between circles is proportional to the differences in energy. Rank the atoms based on the energy of the photon that is absorbed, from lowest to highest. ONLY rank those transitions that absorb a photon and that are physically possible.
Glencoe Physics: Principles and Problems, Student Edition
1st Edition
ISBN:9780078807213
Author:Paul W. Zitzewitz
Publisher:Paul W. Zitzewitz
Chapter28: The Atom
Section: Chapter Questions
Problem 35A
Related questions
Question
- The circles in the diagrams below represent energy levels in an atom, and the arrows show electron transitions from one energy level to another. The spacing between circles is proportional to the differences in energy. Rank the atoms based on the energy of the photon that is absorbed, from lowest to highest. ONLY rank those transitions that absorb a photon and that are physically possible.
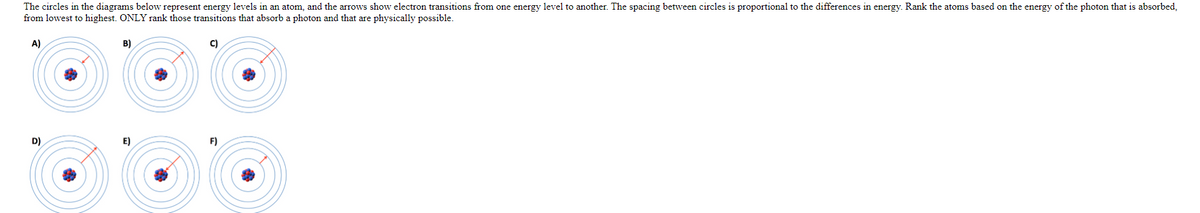
Transcribed Image Text:The circles in the diagrams below represent energy levels in an atom, and the arrows show electron transitions from one energy level to another. The spacing between circles is proportional to the differences in energy. Rank the atoms based on the energy of the photon that is absorbed,
from lowest to highest. ONLY rank those transitions that absorb a photon and that are physically possible.
A)
B)
C)
D)
E)
F)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Glencoe Physics: Principles and Problems, Student…
Physics
ISBN:
9780078807213
Author:
Paul W. Zitzewitz
Publisher:
Glencoe/McGraw-Hill

Modern Physics
Physics
ISBN:
9781111794378
Author:
Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:
Cengage Learning

University Physics Volume 3
Physics
ISBN:
9781938168185
Author:
William Moebs, Jeff Sanny
Publisher:
OpenStax

Glencoe Physics: Principles and Problems, Student…
Physics
ISBN:
9780078807213
Author:
Paul W. Zitzewitz
Publisher:
Glencoe/McGraw-Hill

Modern Physics
Physics
ISBN:
9781111794378
Author:
Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:
Cengage Learning

University Physics Volume 3
Physics
ISBN:
9781938168185
Author:
William Moebs, Jeff Sanny
Publisher:
OpenStax

Foundations of Astronomy (MindTap Course List)
Physics
ISBN:
9781337399920
Author:
Michael A. Seeds, Dana Backman
Publisher:
Cengage Learning

Stars and Galaxies (MindTap Course List)
Physics
ISBN:
9781337399944
Author:
Michael A. Seeds
Publisher:
Cengage Learning
