The concentration of PO43 ions in (M) found in a solution of 3 M H3PO4 is (Ka1= 7.1X10-3, K,2= 6.3X10-8 and Ka3= 4.2X10-13) 6.4 X 10-8 O 5.2 X 10-14 Ο 1.9 Χ 10-19 O 3.7 X 10-23 0.14
Q: Which of the following is an acidic salt?* OKNO3 O SrCl2 O CuCl2 Ba(CH3C00)2
A: Acidic salts are the ones which produce acidic solution when dissolved in solvent. Acidic salts are…
Q: 1. Aqueous dilution of I0, results in the following reaction: 10. jac) + 2H,O H,lO. (ac) and K = 3.5…
A: GIVEN:-
Q: 9 rank the following cations as increasing strength of oxidizing agent : (from weakes to strongest)…
A: In a chemical reaction, the gain of electrons is called reduction and loss of electrons is called…
Q: Which compound listed below will dissolve in carbon tetrachloride, CCl4? OHBr O NaCl O NH3 OBF3 O…
A:
Q: 1. The oxalate ion concentration, C;0. of 0.20 M H;C0 is adjusted to a value of 3.00 x 10 M. What is…
A: As per the guideline, since you have asked multiple questions, we have solved the first question for…
Q: What is the molar concentration of a NH4NO3 solution that has a pH of 6.18? (pKa = 9.25).
A:
Q: The mg/L concentrations of 2.50x104 M solutions of HNO, (F.wt 63 g/mol) equal. 15.57 15.75 17.5 15.5
A:
Q: A solution was prepared by dissolving 2.45 g of AgNO3 in water to give 0.5 liter. Calculate the…
A:
Q: Which of the following is an acidic salt? O NaBr O KNO3 HI O NH4CI O NACIO
A: Acidic salt : strong acid and weak base combination.
Q: What is the activity coefficient of Hg22+ in a solution of 3.3 mM Hg2(NO3)2? O 0.67 O 0.65 O 0.63 O…
A: Given, Concentration of Hg2(NO3)2 is 3.3 mM activity of Hg22+ = ?
Q: 5. Detenmine the moles and number of grams of calcium carbonate needed to make the following…
A: Given (a)25.0ml of 0.997M CaCO3 (b) 1.25L of 3.42M CaCO3 To find moles and mass of CaCO3
Q: NeW August 3, 2021. Describe the preparation of 1.50 L of 0.215 M NaOH from the concentrated…
A: Volume of NaOH = 1.50 L Molarity of NaOH = 0.215 M w/w = 50%
Q: 14. What is the mole ratio of calcium carbonate to carbon dioxide in the reaction between…
A: A mole ratio is a conversion factor that relates the amounts in moles of any two substances in a…
Q: As2. .. What is the minimum pH needed to prepare 1% sodium phenobarbital in water? Intrinsic…
A: We have to prepare 1% sodium phenobarbital in water. Solubility = 0.004M MW = 232.2g/mol pKa = 7.41
Q: What is the concentration of sulfite ion in a 0.100 M sulfurous acid solution? O 1X 10 19 M O 1.0X…
A: Sulfurous acid has chemical formula H2SO3, is a diprotic acid with Ka1 = 1.3 x 10-2 and Ka2 = 6.3 x…
Q: Q5/ What volume of 0.20M Ca(OH)2 will neutralize 45.0 mL of a 1M solution of HCIO;?
A: Given: 0.20 M Ca(OH)2 45.0 mL of 1M HClO3
Q: What is the concentration in % (w/w) of a solution containing 20.0 g of NaCl dissolved in 250.0 g of…
A: Percentage (w/w) means amount of solute divide by amount of solution and multipled by 100.
Q: Barium fluoride (BaF2) has Ksp = 1.8 x 107. VWhat is the maximum fluoride ion concentration possible…
A: Given: Ksp of BaF2 = 1.8 x 10-7 Concentration of Ba2+ = [Ba2+] = 5.0 x 10-4 M
Q: Determine the concentrations of the ionic species present in a 0.403 M solution of the CS2HPO3.…
A:
Q: 33. 0.2347 g of an ionic compound MX (135.32 g/mol) completely dissociates into its corresponding…
A: I request you to pls repost question (37)
Q: Prepare astandared solution of potasstum carbonate K2CO3 2.5M in 200ml of water atomic weight…
A:
Q: The concentration of PO43 ions in (M) found in a solution of 3 M H3PO4 is (Ka1= 7.1X103, K22=…
A:
Q: Part III: An equilibrium system consisting of BiCl3 and bismuth hypochlorite in water was treated…
A: Hello. Since the question contains multiple sub-parts, the first three sub-parts will be solved. If…
Q: Which of the following ionic compounds is INSOLUBLE in water? O LIHCO3. O K2CO2 O LizSO4 Na 2SO 4…
A: The solubility of ionic can be predicted by using solubility rules Some important solubility rules…
Q: You have a solution of KI that is 0.480 M. How would you prepare 5.00 L of a solution that is 1.50x…
A: Given:Molarity of KI = 0.480 M (M1)Volume of required solution = 5.00 L (V2)Conc. Of required…
Q: What is the Charactenshe length of a hydrogen bond m soluhon? a) 0.028 A b) 0. 28 A c) Z.8 Å d) 28 A…
A:
Q: D. The element Radon has the symbol Clź5.452, What is its Atomic Number? How many Neutrons does it…
A: D. The element Radon has the symbol = Rn222 86 , We have to find out Atomic Number and how many…
Q: A solution was prepared by dissolving 2.45, g of Ag2SO4 in water to give 0.5 liter. Calculate the…
A: Given data contains, Mass of Ag2SO4 is 2.45 grams. Volume is 0.5 liter.
Q: What is the concentration of fluoride ions (F) in a solution containing 0.050 M HCI and 0.085 M HF…
A:
Q: dissociate into 3 mole ions
A: Insoluble compounds doesn't dissociate into ions Strong acid fully dissociates into ions
Q: Calculate the concentration of H2SO3 in a 0.510 MM solution of H2SO3 Ka1=1.6×10−2 and Ka2=6.4×10−8).
A: NOTE: In question, you have mentioned 0.510 MM instead of 0.510 M. Hence I'm correcting it and…
Q: Barium fluoride (BaF2) has Ksp = 1.8 x 107. What is the maximum fluoride ion concentration possible,…
A: The maximum fluoride ion concentration can be calculated as follows
Q: Calculate [Sr2+] and [OH−] (in molarity) for a solution that is made by dissolving 2.5 x 10−3 moles…
A:
Q: aqueous solution? Select one: O a. magnesium oxide, MgO O b. hydrogen chloride, HCl O c. dihydrogen…
A: A Bronsted acid is the one which has the ability to donate proton in an aqueous solution.
Q: Calculate the pH of a 0.560 M NaNO2 solution. Ką of HNO2 = 7.1 × 10-4 %3D
A: NaNO2 is a salt of weak acid HNO2 and strong base NaOH. So the aqueous solution of NaNO2 will be…
Q: Sodium methylsulfonate (CH3OSO Na+) is a poor detergent. Explain why. 3.
A: [As per rules only the first question can be answered] A detergent must contain a proper polar part…
Q: 194.3 grams of barium fluoride are weighed out and placed into a 7.82 L volumetric flask and the…
A: Ksp solubility product : when a chemical compound in solid state is in equilibrium with the solution…
Q: What is the concentration of sodium ions in 0.385 M Na₃PO₄?
A: Molarity is a term used for concentration which shows the moles of solute dissolved per liters of…
Q: Sodium tetraphenylborate is an organic precipitating agent for a. Nickel, sodium and ammonium ions.…
A:
Q: What salt is produced in a neutralization reaction between H3PO4 and Mg(OH)2? O MGPO4 O Mg3P2 O…
A: When an acid reacts with the base then a neutralization reaction will occur. In a neutralization…
Q: What is the concentration of calcium ions in a 0.543 M aqueous solution of Ca3P2 O 1.09 M O 0.543 M…
A: The given information is as follows: The concentration of the aqueous solution of Ca3P2 = 0.543 M…
Q: In a 0.072 M solution of chlorous acid, what percentage of the acid is ionized to the chlorous…
A:
Q: 11. What is the pH of 0.05 M NaNO2 solution? [Ka of HNO2 = 4.0 x 10-4] * O 8.0 O 6.0 O 5.0 O 3.4 9.0
A:
Q: Calculate the concentration of all ions present in each of the following solutions of strong ele a.…
A:
Q: Calculate the molarity of 150.0 mL of sodium hydroxide solution needed to react with 65.7 mL of a…
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: What is the maximum molarity of PO43- in a solution that contains 0.20 M Co2+? For Co3(PO4)2 Ksp…
A: Given : Molarity of Co2+ = 0.20 M Co3(PO4)2 Ksp = 2.5 x 10-5
Q: is the number of grams of aluminum sulfate needed to prepare a 0.15 titer solution, 32 = 27 = Al)…
A: Given that - Molarity of the solution = 0.15 M Atomic mass of different elements are - Al = 27 ,…
Q: Identify the oxidizing agent in the following reaction 2Na + 2H2O 2ѪOH + H2 A. Na B. H2O C. NaOH D.…
A: The addition of hydrogen or removal of oxygen is known as reduction and the addition of oxygen and…
21
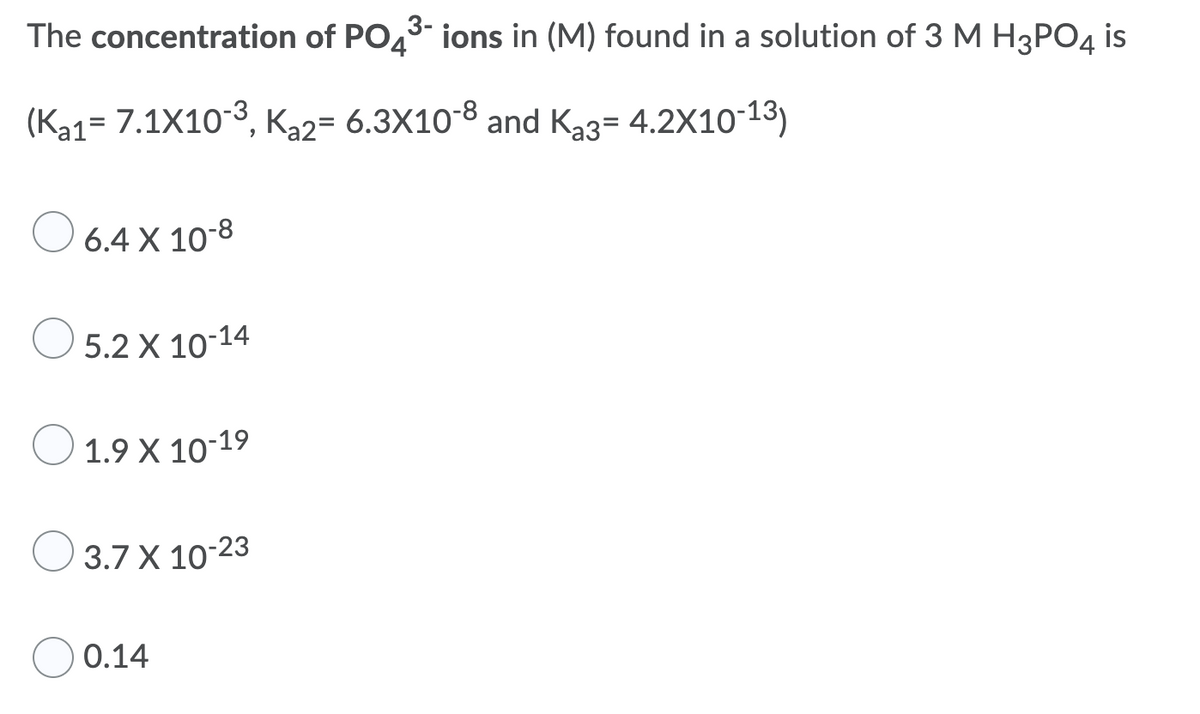
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- A technician is setting up a laboratory to standardise ~0.0200 mol dm–3 KMnO4. What mass of Mohr’s salt [FeSO4(NH4)2SO4·6H2O] would the technician be required to weigh out to produce a 5.00 L solution of Mohr’s salt such that 25.0 cm3 aliquots of the salt would require a titre of 20.0 cm3 to standardise the KMnO4 solution?Hydrogen bromide is a strong electrolyte in water. What is the boiling point elevation of asolution containing 125.0 g of HBr in 1.75 kg of water? Given the following: H= 1,0 g/mol;Br= 79.9 g/mol; boiling point elevation of water is 0.512°C.kg/molA. 1.24 °0B. 101.24 °CC. 0.904 °CD. 100.904 °CGiven: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution (MW: Na: 23, K: 39, Cl: 35, H: 1, C: 12, O: 16) Calculate the total amount of chloride expressed in mmol/L present in the prepared solution 60.34 mmol/L 90.10 mmol/L a 111.11 mmol/L b 29.76 mmol/L c 80.61 mmol/L d 20.27 mmol/L
- If 0.15g of Calcium hydroxide is needed in preparing 50mL of Calcium hydroxide topical solution, calculate the amount of calcium oxide (56g/n) needed in preparing 50 mL of the calcium hydroxide (74g/n) topical solution. CaO + + H2O → Ca(OH)2 reacting ratio: 1 mol CaO = 1mol Ca(OH)2 a 0.20 g b 0.19g c 0.11g d 0.25gPrepare astandared solution of potasstum carbonate K2CO3 2.5M in 200ml of water atomic weight K=39.098 ,O=16 ,C=12Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution (MW: Na: 23, K: 39, Cl: 35, H: 1, C: 12, O: 16) a. Calculate the total amount of glucose expressed in mmol/L present in the prepared solution b. Calculate the total amount of sodium expressed in mmol/L present in the prepared solutionc. c. Calculate the total amount of potassium expressed in mmol/L present in the prepared solution a 111.11 mmol/L b 90.10 mmol/L c 80.61 mmol/L d 60.34 mmol/L e 29.76 mmol/L f 20.27 mmol/L
- How many moles of calcium fluoride (78.07 g/mol) will dissolve in 10.0 L of water at 25°C? Ksp of CaF2 is 3.9 x10-11 0.167 0.00214 2.36 x 10-4 6.24 x 10-6Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution (MW: Na: 23, K: 39, Cl: 35, H: 1, C: 12, O: 16) Calculate the total amount of bicarbonate expressed in mmol/L present in the prepared solution a 90.10 mmol/L b 80.61 mmol/L c 29.76 mmol/L d 60.34 mmol/L e 111.11 mmol/L f 20.27 mmol/LWhat mass of Na3PO4・12H2O would be needed to prepare 0.250L of a solution in which the Na+ ion concentration in 0.250 moles per liter?
- What is the dominant iron species in water samples ? What environmental factors may have contributed. why is it necesseyy to to investigate Fe3+ concentration and total Fe concentration dpeedtaely ? Why do we need to do the sample to determine total Fe concentration ? And how did then determine Fe2+ concentration?Subject :- Chemistry How many grams of EDTA (336.21g/mol) are contained in 1mL of a 2000microM solution?What are the respective concentrations (M) of Fe3+ and I- afforded by dissolving 0.200 mol FeI3 in water and diluting to 725 mL? A) 0.276 and 0.828 B) 0.828 and 0.276 C) 0.276 and 0.276 D) 0.145 and 0.435

