Q: 6 H20) + 6 CO2)- C6H1206(s) + 6 O2), AH-+2803 kl/mol. In order to form 78.155 g of C6H1206 (molar…
A:
Q: In Part A of this experiment, you prepare 5 solutions of FeSCN2* [one of them is just a blank…
A: No of moles = Molarity * Volume (L)
Q: IUPAC name for the following compounds ?
A: The first step in nomenclature is to choose the longest carbon chain which should contain the…
Q: 10. Sulfur trioxide decomposes according to the reaction: 2 SO3 (g) = 2 S02 (g) + O2 (g) %3D In an…
A:
Q: d. Terbutaline 25. Which of the following agents can enhance the sedative action of promethazine…
A:
Q: Draw the structures of compounds M and N NaOEt HBr M N ELOH
A:
Q: T Write out the mechanism for the following reaction. Show all steps and use 1. curved arrows. H20 +…
A: Given,
Q: The specific heat capacity of liquid ammonia is 4700J/Kg.K and its latent heat of vaporization is…
A:
Q: how to determine the formula for the complex copper(II) ammine ion?
A:
Q: 7. CH,O, + OH CH,O,+ H,O redox reaction (yes/no), species oxidized oxidizing agent species reduced…
A: 7. C2H4O2 + OH- -----> C2H3O2- + H2O Here the oxidation states of different atoms at reactant…
Q: If you are given moles and liters, you would be able to solve for (without using any conversion…
A:
Q: What is a standardization and why is it important?
A: Concept: Standardization: Standardization is the process of determining the exact concentration of…
Q: Given the rate constant, k, for the decay of a radioactive substance, how would you find the…
A: Given :- rate constant for decay of radioactive substance = k To find :- half-life (t1/2)
Q: Which substances in the equation below are classified as Bronsted-Lowry ACIDS: H2S (aq) + H2O0 → H30…
A:
Q: Consider the following reaction (DPPE structure below): F. - B(OH)2 F Ni(OTf)2 DPPE K2CO3 (a) Give…
A: The name reaction of the given transmetalation is known as Suzuki-Miyaura coupling in which metal…
Q: Consider a 1:1 ligand-receptor binding interaction modeled by X + Y 2 XY, where X is the ligand, and…
A:
Q: Write a balanced nuclear equation for this word statement. Magnesium-28 undergoes beta emission.
A:
Q: Balance the given redox reactions using the oxidation number method a.H2S(aq) + HNO3 (aq) -> S(s) +…
A: Oxidation number is the hypothetical charge of an atom if all its bonds to different atoms are fully…
Q: A 24.0-liter gas cylinder contains 8.0 moles of nitrogen gas and 2.3 moles of oxygen gas. If the…
A: Given :- No.of moles of O2 = 2.3 mol No.of moles of N2 = 8.0 mol Volume = 24.0 L Temperature =…
Q: 25. Which of the following is usually considered to be a weak acid? A) HCI B) HBr C) HI D) HF 26.…
A: Using formula: PH = - log[H+] where as [H+] --->> concentration of H+ ion
Q: You have synthesized Below compounds: 1 K[Co2(H,O)(CN),Br) 2 [Mn,(H,O)(CN),CI,JSO, Find If there is…
A: Hello dear, welcome on bartleby; It is the problem of organometallic chemistry and the detailed…
Q: Use the space provided below to draw a flow chart showing how you could separate a mixture…
A: The flow chart for the extraction of given compounds has been shown below:
Q: Ethyl acetate (C4H8O2, mm=D 88.10 g/mol) is a colorless liquid often used in nail polish removers,…
A: ∆H°vap of ethyl acetate is 31.94 kJ/mol. Kb for ethyl acetate is 2.77 °C kg/mol. It is required to…
Q: Given the following sets of initial conditions, what is the net change that must occur for the…
A:
Q: 41. Given the following Ka values, which anion is the strongest base? Acid H,CO; HCO;- H,C,04 HC;O,…
A: For the given Ka values, which anion is strongest among them---
Q: +2 a. Agac + 2NH3 (acy Ag (NH3)2 (ac) b. AgCls) + Agac)+ Clac) Ni(s) + 4CO) c. Ni(CO)4 (0) * d. CH3…
A: Below attached file showing the details answer. All the best.
Q: xsH20 - A CH2=CH-SIMECI2 heating xsH2O HSİMECI2 B H2SO4
A: A and B formed by hydrolysis of Si-Cl bond in presence of excess water. Then the A and B condensed…
Q: with a weak acid, either HA, HB. HC. or HD. Rank the acids HA, HB, HC, and HD in order from…
A:
Q: Write the palladium-containing product for the following reaction and indicate its stereochemistry:…
A: We can solve this question by using the concept of trans effect where CN- regulates the substitution…
Q: QUESTION 41 Which of the following solutions is a good buffer system? A solution that is 0.10 M HI…
A:
Q: Predict the major product(s) obtained the following compound undergoes hydrolysis in the presence of…
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please…
Q: A solution is prepared by combining 20.0 mLs of 0.10 M solution of an unknown acid and 15.0 mLs of…
A:
Q: If 1.50 mL of a stock solution of 12.0 M HCl is diluted to 25.0 mL, what is the new concentration?
A:
Q: 4 Define the following symbols: a. S d. G b. AS e. AG с. Т
A:
Q: 39) Complete the multi-step synthesis by showing the product structure of each step. NaNH2 H/Pd CH…
A:
Q: Match the line structure to its corresponding structural formula. 1. HHHH H H-C-C-C-C-C-OH HHHH H 2.…
A:
Q: What is the enthalpy of the reaction, in kJ/mol, knowing that 7.5 g KNO3 absorbs 2597 J of energy?…
A:
Q: Zn (s) + 2HBr (aq) --> H2 (g) + ZnBr2 (aq) What is the molarity of an HBr solution if the…
A: Given :- Zn (s) + 2HBr (aq) --> H2 (g) + ZnBr2 (aq) Volume of H2 = 0.620 L Pressure of H2 =…
Q: Chemistry 5. Write the notation for the following cells where the following reactions occur…
A: To write cell notation for the following cells given
Q: How many atoms of hydrogen are found in 3.92 mol of hexane, C6H14?
A:
Q: According to the following reaction, how many grams of nitrogen gas will be formed upon the complete…
A: Given Mass of NH4NO2 = 28.0 g We know that, Number of mol = mass / molar mass Number of mol of…
Q: Current Attempt in Progress What is the mass defect for americium-241 if the mass of the nucleus is…
A:
Q: 33. Nickel crystallizes in an fcc structure, with a unit cell edge length of 352 pm. What is the…
A: Concept: Relation between radius of atom and edge length: The relation between edge length (a) and…
Q: Draw the skeletal (line-bond) structure of (Z)-2-ethyl-N-methylhept-2-enamide. Organized Please.
A: IUPAC nomenclature is used for naming the organic compound as recommended by international union of…
Q: Question31 Which of the following partially soluble compounds would not be more soluble in acidic…
A: Concept: salt are Three types 1. Acidic salt: They are formed by reaction of weak base and strong…
Q: A 1.22 gram sample of an unknown gas is found to occupy a volume of 1.13 L at a pressure of 441 mmHg…
A:
Q: What is the formula for the complex ion made by iron(IIl) with 4 aqua and two thiosulfato (S,0022)…
A: Known Metal ion - Fe3+ ; ligands are 4 aqua - 4 H2O S2O32- Complex should be written in square…
Q: 30) Show an increase in the order of the acidity for the following aromatic acids: (i) p-ethyl…
A: Acidity of benzoic acid depends on the group that attached to the benzene ring and the position of…
Q: The sketches are drawn as if a sample of carbon monaxide were under a microscope so powerful that…
A: Solution - The correct option is sketch (C) Explanation -
Q: Calculate e values for the galvanic cells based on the following overall reactions. Assume that all…
A: The given reactions are a. Cr3+(aq) + Cl2(g) ⇔ Cr2O72-(aq) + Cl-(aq); E0cell = V b. Pb2+(aq) + Ba(s)…
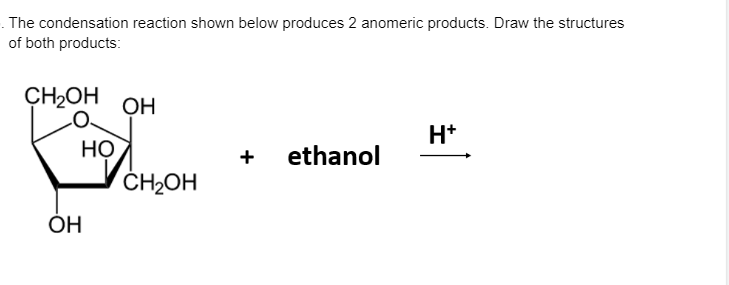
Carbohydrates
Step by step
Solved in 2 steps with 2 images

- Draw the structure of the product of the reaction shown above. (Take Note: Sia2BH and H2O2, NaOH is used)What product would occur after an addition reaction of H2O to the following acid? How would the properties of the new product be different from the original acid?what is the expected major product of HBr addition to the alkene shown below? Show the mechanism
- Name the following alkene and draw the product of the reaction with KMnO4.For the dehydration shown, use curved arrows to show the formation of the carbocation intermediate in the presence of sulfuric acid H2SO4H2SO4, then draw the structures of the minor and major products of the elimination.Predict the major products formed when 2- methyl-1-butene reacts with: H2O, H+. Show the reaction mechanism of the given alkene reactions
- Show the organic product formed when 4-methylpentanal is heated with sodium methoxide in methanol as solvent. Show the steps in the mechanism as reactant is converted to final product. show appropriate arrow pushing and any charges,and draw a box around the final product.In the scheme below, different products are obtained with the reagents given starting from 2-methyl-3-hydroxypentanal. Write the open structures of the products D E, F, G.Show the products generated from the HBr reaction with each of the following alkenes, not forgetting to indicate which is the majority product.
- Hydrogenation of 3-carene (below) can yield two steroisomeric alkane products. Draw their structures. The reaction actually only yields one of the two products. Put a box around the product formed and explain your choice.The substitution reaction below forms two ehters (which are consitutional isomers). Please draw the structures of the 2 ethers formed. Are the ethers single enantiomers or racemic mixtures?Consider the reaction given below. What are the major products expected from the reaction of this molecule? Show all the possible MAJOR products including stereoisomers. Show the intermediate leading to each product.

