Q: Vd increases, t1/2 ______ because____ A. Decrease; t1/2 proportional to 1/Vd B increase; t1/2…
A: Vd is Directly proportion to the half life that is t1/2 So according to this above statement we can…
Q: by moving bound argy expeciment fos C1N kd, calculate the jonic mobitity enf kt jf applied potendial…
A:
Q: Calculate the pK at 25 degrees C of the following reaction using the data in the table. Use R =…
A: The given reaction is:4 A + 2 B → C + 3 D + 4…
Q: Which of these following positions has the highest value of E°? B D 1.297 V -0.05 V NO3 0.803 V N204…
A: Latimer diagram :- A Latimer diagram of a chemical species is a summary of the standard electrode…
Q: 19 19 Using the information given below, calculate ASuniy for the following rxn at 298 K. 2 C,H2(g)…
A: ∆S(univ) = ∆S(system) + ∆S(surrounding).
Q: the Ksp expression of: Write @ Ag Noz (s) b) Ag2 Sou (s)
A: Given-> (1)->AgNO2 (2)->Ag2SO4
Q: Calculate ΔH°rxn using the following information: SO2I2(g) + 2 H2O(l) → 2 HI(g)…
A: For the given reaction: SO2I2(g) + 2 H2O(l) → 2 HI(g) + H2SO4(l) ΔH°rxn for the above given…
Q: The Ka for HC2H3O2 is 1.8 x 10-5. What is the value of Kb for C2H3O2?
A: Given, Ka for HC2H3O2 is 1.8 x 10-5
Q: find deltaE q = 5.700 kJ; w = 0.8000 J
A: Given that,: q = 5.700KJ w = 0.8000J = 0.8000J×1KJ/1000J = 0.0008 KJ
Q: Determine the values of Kc that correspond to the following values of Kp. 1.)…
A: The relationship between Kc and Kp follows: Kp=Kc RT∆ng .....(1) where, Kp =…
Q: Fimd मरe vabue ofीर स खेड भमेंग्टन the value भेप्ज baleoeeaction CaHy t 402- > 3 C02+2t2O -1790KJ…
A: Given reaction is : C3H4 + 4O2 ----------> 3CO2 + 2H2O Calculate the standard change in…
Q: Calculate for the solubility of PbF2, given the value of its Ksp. Ksp of PbF2 = 3.3 x 10-8
A:
Q: For each of the following molecules and ions, identify if it has a centre of inversion (i) and/or an…
A: It is a type of symmetry in Inorganic molecules. Center of inversion means all other atoms can…
Q: Calculate the pK at 25 degrees C of the following reaction using the data in the table. Use R =…
A: The difference between the initial-state energy and the equilibrium-state energy is known as free…
Q: 1000 500 250 400 300 200 100 Ó CPS 1H NMR 60 ΜHz 200 50 100 80 Integral = 3 50 Integral = 3 Integral…
A: Double bond equivalence value is given by, DBE= (no of Carbon +1) - monovalent atom/2 + trivalent…
Q: he partial character table of D4h is given below. E 2C, (2) C2 2C'2 2c", i 25, on 20, 204 2S4 on 20,…
A: Solution -
Q: NI = 12037 N2 d2= 9.24x1o 6 year! iニ 2.83 X 106 yeari! You have to determòne t value from the…
A:
Q: Calculate the pK at 25 degrees C of the following reaction using the data in the table. Use R =…
A:
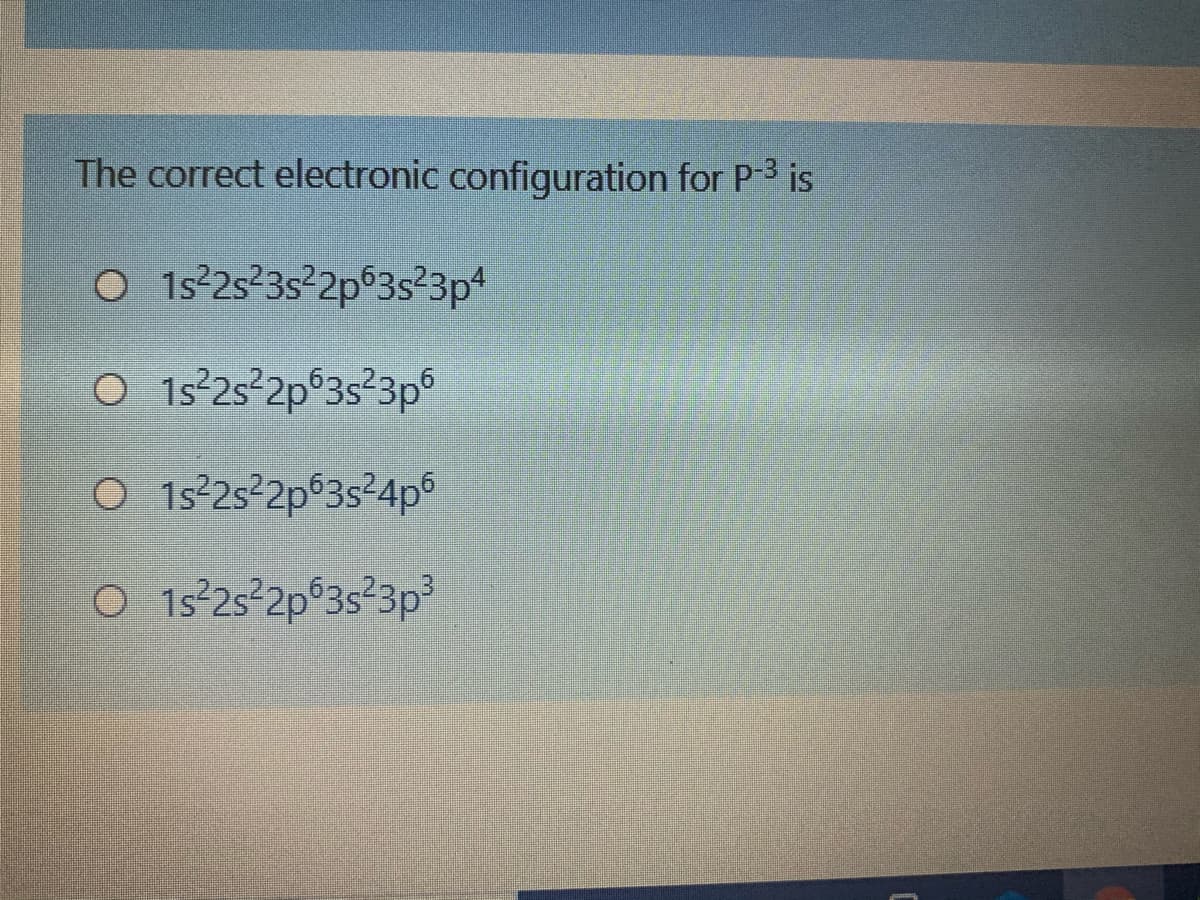
Q: on the right. 1s 2s 2p 3s 3p 4s²3d¹04p5s²4d¹05p 1s 2s 2p 3s 3p 4s²3d¹04p5s²4d¹05p6s 1s 2s 2p 3s 3p…
A: The electrons which are present in the outermost shell are called the valence electrons. Normally…
Q: 2 13 14 15 16 17 googlegalaxyaclence.com 2-1 Li Be B N F 1-0 1-5 2-0 2-5 3-0 3-5 4. Na 0-9 Si P Mg…
A: Solution- by the observed, We found as across in given periodic table in the 1st line…
Q: Consider the following equation that is commonily used in pharmacokinetic studies of drugs In[D]e =…
A: To solve this problem first we have to rearrange the given equation to get K
Q: Find the percent error CACO3(s)= -1206.9 KJ/mol CaO(s)= -635.5 KJ/mol CO2(g)= -393.5 KJ/mol (AH°;)…
A: Given that : We have to calculate the % error :
Q: MCQ 22: The name given to sub-shells are s-sharp, p-principal and d for A. different B. diffuse C.…
A: The given options are,
Q: Н (еxcess) O,N-
A: Degree of unsaturation is analysis of structure of an organic molecule. It helps in determining the…
Q: s, 9H s, 2H s, IH 12 10 8 4 2 -2 HSP-03-150 ppm но. O 1.A O 2. B O 3. C O 4.D O 5. E
A: The number of peaks in 1H NMR indicates number of nonequivalent protons present the given organic…
Q: concentration
A: Given Details: Weight of Molecular = 84180 g/mol concentration = 0.04g/cm3 molecular weight = 28…
Q: Provided the following information determine ΔHformation of C2H6(g). C(graphite) + O2(g) ⟶…
A:
Q: Dotougite the Cnplex frans((oil2 (usl3)) qnd Complex trans-twifrntB) THust vatet 0Ptilalisamers…
A: coordination compounds are generally tetrahedral, spqare planar and octahedral. bonding direction of…
Q: Consider the following arbitrary compound and choose as to which statement is correct about it: PDF:…
A: Effect of electronegative element on bond breaking: Electronegative element have tendency to…
Q: Which of the following is true for a nonspontaneous process? (d is for delta) 1.dH>TdS 2.dH<TdS…
A: Which of the following is true for a nonspontaneous process? (d is for delta) 1.dH>TdS…
Q: Calculate delta H for the graph below. y = -59.88x -1.6547 -1.84 -1.645 -1.85 1.800 Ink vs 1/T…
A: The equation for a linear graph is given by y=mx + c where m is the slope and c is the y-intercept.
Q: Bb MYJCC Blackboard Learn WebAdvisor for Students JCC Self Service Bb UTF-8 Syllabus_CHM%20151%2 X A…
A: As per regulations, only three sub-parts can be answered.
Q: Define the Hückel's Rule ?
A: The compounds containing carbon and hydrogen are termed as hydrocarbons. Hydrocarbons are classified…
Q: Carbon has electronic configuration 1s2 2s2 2p2 . How will you justify its tetravalency?
A: Step : 1 Carbon has electronic configuration 1s2 2s2 2p2 . How will you justify its…
Q: exe/lo_u-lgNsikr7j8P3jH-IQs_dp5pR4ENzvdYC-70kXyMz36BqJhw3sVPj_jpaFLxvGArYxlbmcyqa71YYPJBG6RjdYAdGPjG…
A: The rate constant for a certain reaction is known to obey the Arrhenius equation, and have an…
Q: 39) Give the complete electronic configuration for Mn. A) 1s22522p63s23p64s23d5 C)…
A: We have to give electronic configuration of Mn
Q: Please calculate the energy of inward movement of chloride ions given: Cl- F=23100 cal/V*mol Vm =…
A: Given information, F=23100 cal/V*mol Vm = -60mV R =2cal/mol*K T = 300K Inside 50mM Outside 150mM z…
Q: K2Cr207
A:
Q: 2. Calculate the FW/MW of the ff; NH3 b. CaCl2 а. c NaCIO3 С. d. Ba(OH)2 e. Саз (РО4)2 е.
A: We have to calculate the MW of the given compounds as follows in step 2:
Q: Calculate AG" (In kj/mol) given AG- 451.5 kl/mol and R 0.008314 kj/mol K and T 854.8 K and Q-0.799…
A: The correct answer is given below
Q: 3, "The DeNo) valurin [ Ru (up), (ni) x]" are x - MecN, z:3, VNo = (970 cm!! X=cl, Z=2, VCNO1927 cm?…
A: Electrophilic behaviour is dependent upon IR Streching Frequency which is explained below:
Q: ng point determination can be used for several purposes. What are those purposes? effect on the…
A: Condensation is the change of vapour into liquid and evaporation is the change of liquid into…
Q: Identify the atom oxidized, the atom reduced, the oxidizing agent, and the reducing agent
A: Dear student I have given answer to first three subparts of your question in the image format. If…
Q: 1, CH3MGB1, Et2O 2. H", Н.О Selected Coordinates Clear
A:
Q: 4) Use the information below to find AHpn for... 2MNO2(aq) + CO(g) → Mn2O3(aq) + COz(g) Using the…
A: The final equation is obtained by the following relation: ∆H0rxn= (1/3) ×3 + (2/3) ×2 – 2 ×1
Q: 1) 4) O R 2) 8 5) AcO 3) p 6) c∞
A: UV- Visible Spectroscopy : UV - Visible spectroscopy is an anlaytical technique where the…
Q: Using the information given below, calculate ASv for the following rxn at 298 K. sys Al,O3(s) +…
A: "Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: 36R) Which one of the following configurations depicts an excited oxygen at A) 1s22s22p2 B)…
A: Oxygen atom having atomic number 8 having 8 electrons.
Q: Given that = -546.6kJ H,(g) +F,(g)→ 2 HF(g) rxn AHixn = -571.6 kJ (1DO H7 - (8) o+ (8) H calculate…
A:
Step by step
Solved in 2 steps with 1 images

- ᴀᴄᴛɪᴠɪᴛʏ ₃: ʀᴇᴀᴄᴛɪᴏɴꜱ ᴏʀ ᴛᴇᴄʜɴɪQᴜᴇꜱ ɪɴ ʏɪᴇʟᴅɪɴɢ ᴏᴄ!ᴅɪʀᴇᴄᴛɪᴏɴꜱ: ɪᴅᴇɴᴛɪꜰʏ ᴛʜᴇ ʀᴇᴀᴄᴛɪᴏɴ ᴏʀ ᴘʀᴏᴄᴇꜱꜱ ʙᴇɪɴɢ ᴅᴇꜱᴄʀɪʙᴇᴅ._________________ ₁. ɪᴛ ɪꜱ ᴀ ᴍᴇᴛᴀʙᴏʟɪᴄ ᴘʀᴏᴄᴇꜱꜱ ɪɴ ᴡʜɪᴄʜ ᴀɴ ᴏʀɢᴀɴɪꜱᴍ ᴄᴏɴᴠᴇʀᴛꜱ ᴀᴄᴀʀʙᴏʜʏᴅʀᴀᴛᴇ, ꜱᴜᴄʜ ᴀꜱ ꜱᴛᴀʀᴄʜ ᴏʀ ᴀ ꜱᴜɢᴀʀ, ɪɴᴛᴏ ᴀɴ ᴀʟᴄᴏʜᴏʟ ᴏʀᴀɴ ᴀᴄɪᴅ. _________________ ₂. ᴛʜɪꜱ ʀᴇᴀᴄᴛɪᴏɴ ɪꜱ ᴅᴏɴᴇ ᴛʜʀᴏᴜɢʜ ʀᴇᴍᴏᴠᴀʟ ᴏꜰ ᴡᴀᴛᴇʀ ᴀɴᴅ ᴍᴀʏ ʏɪᴇʟᴅ ᴀɴ ᴀʟᴋᴇɴᴇ ᴀɴᴅ ᴀ ᴍᴏʟᴇᴄᴜʟᴇ ᴏꜰ ᴡᴀᴛᴇʀ. _________________ ₃. ɪᴛ ɪꜱ ᴀ ᴘʀᴏᴄᴇꜱꜱ ʙʏ ᴡʜɪᴄʜ ᴄᴏᴍᴘᴏɴᴇɴᴛꜱ ɪɴ ᴀ ᴄʜᴇᴍɪᴄᴀʟ ᴍɪxᴛᴜʀᴇ ᴀʀᴇꜱᴇᴘᴀʀᴀᴛᴇᴅ ɪɴᴛᴏ ᴅɪꜰꜰᴇʀᴇɴᴛ ᴘᴀʀᴛꜱ (ᴄᴀʟʟᴇᴅ ꜰʀᴀᴄᴛɪᴏɴꜱ) ᴀᴄᴄᴏʀᴅɪɴɢ ᴛᴏᴛʜᴇɪʀ ᴅɪꜰꜰᴇʀᴇɴᴛ ʙᴏɪʟɪɴɢ ᴘᴏɪɴᴛꜱ. _________________ ₄. ɪɴ ʟᴀʙᴏʀᴀᴛᴏʀʏ, ɪᴛ ɪꜱ ᴜꜱᴜᴀʟʟʏ ᴄᴀʀʀɪᴇᴅ ᴏᴜᴛ ᴡɪᴛʜ ᴀᴄᴇᴛʏʟ ᴀɴʜʏᴅʀɪᴅᴇ._________________ ₅. ɪᴛ ɪꜱ ʀᴇᴀᴄᴛɪᴏɴ ᴛʜᴀᴛ ɪꜱ ᴜꜱᴇᴅ ᴛᴏ ᴏʙᴛᴀɪɴ ᴇꜱᴛᴇʀꜱ ʙʏ ᴛʜᴇ ʜʏᴅʀᴏxʏʟ ɢʀᴏᴜᴘᴏꜰ ᴛʜᴇ ᴄᴀʀʙᴏxʏʟɪᴄ ᴀᴄɪᴅ ᴀᴄᴛꜱ ᴀꜱ ᴀ ʟᴇᴀᴠɪɴɢ ɢʀᴏᴜᴘ ᴀɴᴅ ꜰᴏʀᴍꜱ ᴛʜᴇᴡᴀᴛᴇʀ ᴍᴏʟᴇᴄᴜʟᴇ ɪɴ ᴛʜᴇ ꜰɪɴᴀʟ ᴘʀᴏᴅᴜᴄᴛ. ɪᴛ ɪꜱ ʀᴇᴘʟᴀᴄᴇᴅ ʙʏ ᴛʜᴇ -ᴏʀɢʀᴏᴜᴘ ꜰʀᴏᴍ ᴛʜᴇ ᴀʟᴄᴏʜᴏʟ.You can use this as your reference: https://www.youtube.com/watch?v=2J2t5FRnMtMWhich of the following are not allowed? State which rule is violated for any that are not allowed. (a) 1p3 (b) 2p8 (c) 3g11 (d) 4f2
- 17.46 parts d,e,fF i n d t h e p H a t e qu i v a l en c e po i n t i f : A 5 0 m L o f 0 . 1 2 0 M H C N (K a H C N = 6 . 2 0 x 1 0 - 1 0 ) I s t it r a te d w i t h 0 . 1 0 0 M N a O H.Chemistry (i) For T = 298K ΔG° = -8.314 x 298 x ln (2.83x10-3) = 14.524 KJ/mol (ii) For T = 308.15K ΔG° = -8.314 x 308.15 x ln (9.619x10-3) = 11.891 KJ/mol (iii) For T = 318.15K ΔG° = -8.314 x 318.15 x ln (3.14x10-2) = 9.147 KJ/mol (iv) For T = 328.15K ΔG° = -8.314 x 328.15 x ln (8.82x10-2) = 6.621 KJ/mol (v) For T = 338.15K ΔG° = -8.314 x 338.15 x ln (2.42x10-1) = 3.990 KJ/mol Based on the measured ΔG° values, is this equilibrium spontaneous at room temperature? Which factor, entropy, or enthalpy, has the greater impact on spontaneity in this case? Explain your answers.

