The correct value according to Ruler A is 2.2 cm, whereas the correct value according to Ruler B is 2.27 cm. Perform the mathematical operations indicated by the title of each bin using these two values. Round the correct answers to the appropriate number of significant figures or decimal places. Compare the rounded correct answers with the values given and drag the rounded correct answers to the bin that represents the mathematical operation performed. There are also answers that are not rounded to the correct number of significant figures or decimal places. Drag these answers to the bin named "Incorrect rounding." For example, multiply the number 2.2 by 2.27. Round the answer to the appropriate number of significant figures. Drag the correctly rounded answer to the bin labeled "2.2 x 2.27." Drag the incorrect answers from the multiplication operation to the bin named "Incorrect rounding." Drag the appropriate items to their respective bins. • View Available Hint(s) Reset Help 4.994 cm? 5.0 cm? 1.0 0.1 cm 0.07 cm 1 4.47 cm 4.99 cm? 1.03 4.5 cm 0.0 cm 2.27 x 2.2 =? 2.27/2.2 =? 2.27 +2.2 = ? 2.27 – 2.2 = ? Incorrect rounding
The correct value according to Ruler A is 2.2 cm, whereas the correct value according to Ruler B is 2.27 cm. Perform the mathematical operations indicated by the title of each bin using these two values. Round the correct answers to the appropriate number of significant figures or decimal places. Compare the rounded correct answers with the values given and drag the rounded correct answers to the bin that represents the mathematical operation performed. There are also answers that are not rounded to the correct number of significant figures or decimal places. Drag these answers to the bin named "Incorrect rounding." For example, multiply the number 2.2 by 2.27. Round the answer to the appropriate number of significant figures. Drag the correctly rounded answer to the bin labeled "2.2 x 2.27." Drag the incorrect answers from the multiplication operation to the bin named "Incorrect rounding." Drag the appropriate items to their respective bins. • View Available Hint(s) Reset Help 4.994 cm? 5.0 cm? 1.0 0.1 cm 0.07 cm 1 4.47 cm 4.99 cm? 1.03 4.5 cm 0.0 cm 2.27 x 2.2 =? 2.27/2.2 =? 2.27 +2.2 = ? 2.27 – 2.2 = ? Incorrect rounding
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 34E: The beakers shown below have different precisions. a. Label the amount of water in each of the three...
Related questions
Question
Please answer question 12 part B

Transcribed Image Text:Rules for rounding off answers in calculations
While performing a mathematical operation on numbers that vary in significant figures, you follow certain rules. These rules are as follows:
1. For multiplication or division, the answer will have the same number of significant figures as that of the number that has the fewest significant figures.
For example, when multiplying 1.2, 1.58, and 1.222, the product will be equal to 1.2 × 1.58 × 1.222 = 2.3169. However, this number will be rounded off to two significant
figures because 1.2 has the fewest significant figures. Thus, the product will be 2.3.
2. For addition or subtraction, the answer will have the same number of decimal places as that of the number with the fewest decimal places.
For example, when adding 1.2, 1.58, and 1.222, the sum will be equal to 1.2 + 1.58 +1.222 = 4.002. However, this number will be rounded off to 4.0, as 1.2 is the
number with the fewest decimal places.
Part B
The correct value according to Ruler A is 2.2 cm, whereas the correct value according to Ruler B is 2.27 cm. Perform the mathematical operations indicated by the title of each bin
using these two values. Round the correct answers to the appropriate number of significant figures or decimal places. Compare the rounded correct answers with the values given and
drag the rounded correct answers to the bin that represents the mathematical operation performed. There are also answers that are not rounded to the correct number of significant
figures or decimal places. Drag these answers to the bin named "Incorrect rounding." For example, multiply the number 2.2 by 2.27. Round the answer to the appropriate number of
significant figures. Drag the correctly rounded answer to the bin labeled "2.2 x 2.27."
Drag the incorrect answers from the multiplication operation to the bin named "Incorrect rounding."
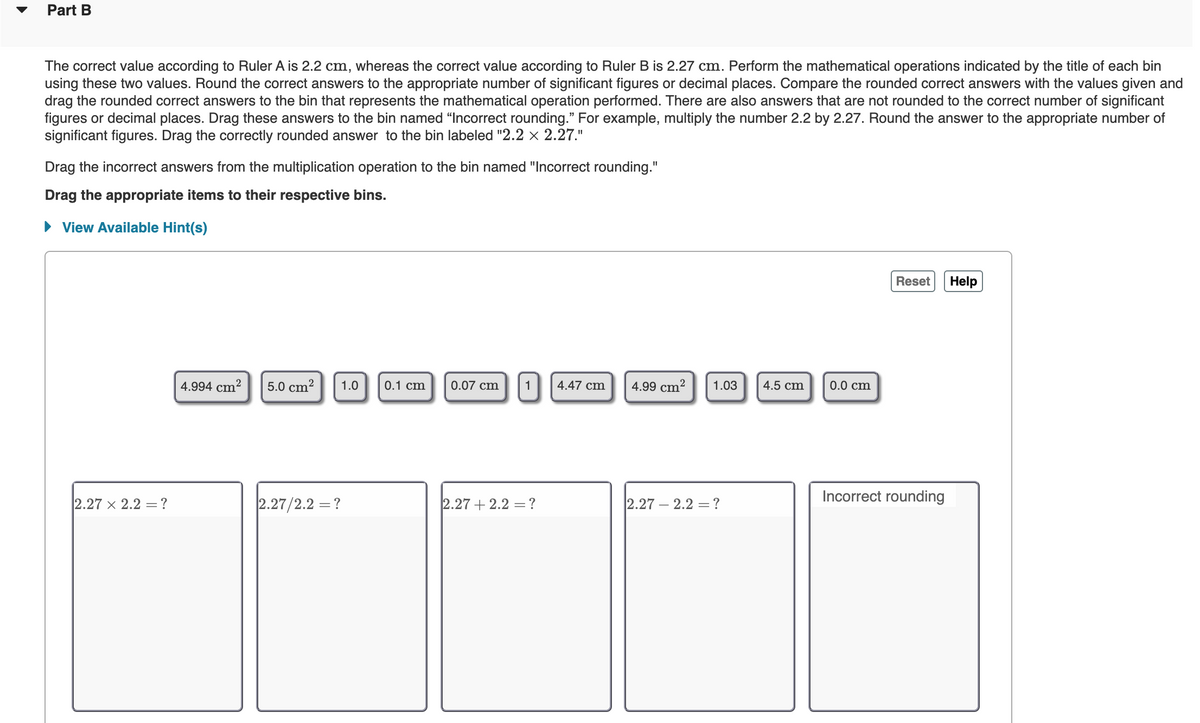
Transcribed Image Text:Part B
The correct value according to Ruler A is 2.2 cm, whereas the correct value according to Ruler B is 2.27 cm. Perform the mathematical operations indicated by the title of each bin
using these two values. Round the correct answers to the appropriate number of significant figures or decimal places. Compare the rounded correct answers with the values given and
drag the rounded correct answers to the bin that represents the mathematical operation performed. There are also answers that are not rounded to the correct number of significant
figures or decimal places. Drag these answers to the bin named "Incorrect rounding." For example, multiply the number 2.2 by 2.27. Round the answer to the appropriate number of
significant figures. Drag the correctly rounded answer to the bin labeled "2.2 × 2.27."
Drag the incorrect answers from the multiplication operation to the bin named "Incorrect rounding."
Drag the appropriate items to their respective bins.
• View Available Hint(s)
Reset
Help
0.1 cm
1
0.0 cm
4.994 cm?
5.0 cm?
1.0
0.07 cm
4.47 cm
4.99 cm?
1.03
4.5 cm
Incorrect rounding
2.27 x 2.2 =?
2.27/2.2 = ?
2.27 + 2.2 =?
2.27 – 2.2 = ?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning