The decomposition of N2O5 when heated in carbon tetrachloride solvent is a second order reaction. 2N2O5(g) → 4NO2(g) + O2(g) The rate constant for the decomposition reaction is 8.1×10-4 M-1s-1 at 350K. What is the half-life in hours for the reaction at 350 K if the initial concentration of N2O5 is 0.0550 M?
The decomposition of N2O5 when heated in carbon tetrachloride solvent is a second order reaction. 2N2O5(g) → 4NO2(g) + O2(g) The rate constant for the decomposition reaction is 8.1×10-4 M-1s-1 at 350K. What is the half-life in hours for the reaction at 350 K if the initial concentration of N2O5 is 0.0550 M?
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter11: Rate Of Reaction
Section: Chapter Questions
Problem 39QAP: The decomposition of azomethane, (CH3)2N2, to nitrogen and ethane gases is a first-order reaction,...
Related questions
Question
The decomposition of N2O5 when heated in carbon tetrachloride solvent is a second order reaction.
2N2O5(g) → 4NO2(g) + O2(g)
The rate constant for the decomposition reaction is 8.1×10-4 M-1s-1 at 350K. What is the half-life in hours for the reaction at 350 K if the initial concentration of N2O5 is 0.0550 M?
Expert Solution
Step 1
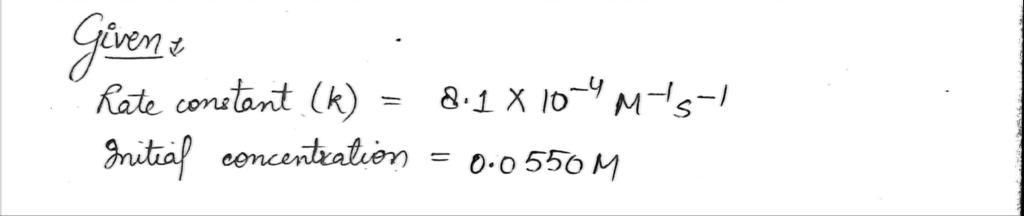
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning