The decomposition of NOCI (g) to NO (g) and Cl₂ (g) was executed in a reactor at 500 K wherein 4.98 moles of NOCI was placed in a 1.50-liter reactor until the system reached equilibrium. At equilibrium, the contents of the reactor were found to contain 0.54 moles of Cl2. Compute for the equilibrium constant Kat 500 K. Express your answer in three decimal places. 2NOCI (g) + 2NO(g) + Cl₂ (g)
The decomposition of NOCI (g) to NO (g) and Cl₂ (g) was executed in a reactor at 500 K wherein 4.98 moles of NOCI was placed in a 1.50-liter reactor until the system reached equilibrium. At equilibrium, the contents of the reactor were found to contain 0.54 moles of Cl2. Compute for the equilibrium constant Kat 500 K. Express your answer in three decimal places. 2NOCI (g) + 2NO(g) + Cl₂ (g)
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter14: Chemical Equilibrium
Section: Chapter Questions
Problem 21P
Related questions
Question
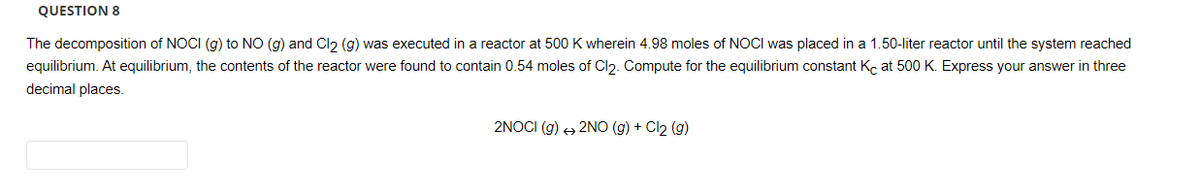
Transcribed Image Text:QUESTION 8
The decomposition of NOCI (g) to NO (g) and Cl2 (g) was executed in a reactor at 500 K wherein 4.98 moles of NOCI was placed in a 1.50-liter reactor until the system reached
equilibrium. At equilibrium, the contents of the reactor were found to contain 0.54 moles of Cl₂. Compute for the equilibrium constant Kc at 500 K. Express your answer in three
decimal places.
2NOCI (g) + 2NO(g) + Cl₂ (g)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning