The development of the Haber-Bosch process for manufacturing ammonia from elemental hydrogen and nitrogen was a major industrial revolution that had a significant effect during World War I. To maximize the percent yield, the reaction is carried out at 20.0 MPa and 450 °C. Calculate the percent yield if 750 L of ammonia gas is collected when 21.8 kg of hydrogen gas is reacted with excess nitrogen gas. Hint: Write a balanced chemical equation first. Do not show your work in the space provided. Record only your final answer with the correct number of significant digits and the proper units. Answer:
The development of the Haber-Bosch process for manufacturing ammonia from elemental hydrogen and nitrogen was a major industrial revolution that had a significant effect during World War I. To maximize the percent yield, the reaction is carried out at 20.0 MPa and 450 °C. Calculate the percent yield if 750 L of ammonia gas is collected when 21.8 kg of hydrogen gas is reacted with excess nitrogen gas. Hint: Write a balanced chemical equation first. Do not show your work in the space provided. Record only your final answer with the correct number of significant digits and the proper units. Answer:
Chapter5: Gases
Section: Chapter Questions
Problem 74E: Urea (H2NCONH2) is used extensively as a nitrogen source in fertilizers. It is produced commercially...
Related questions
Question
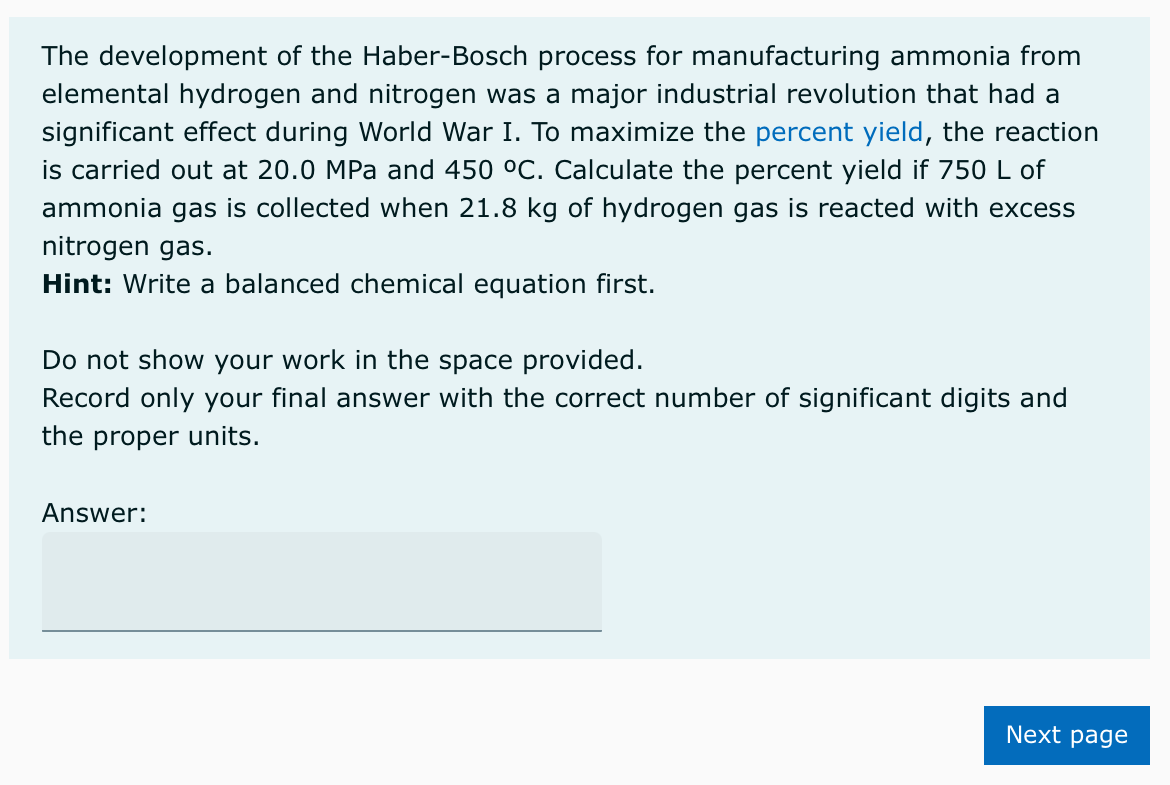
Transcribed Image Text:The development of the Haber-Bosch process for manufacturing ammonia from
elemental hydrogen and nitrogen was a major industrial revolution that had a
significant effect during World War I. To maximize the percent yield, the reaction
is carried out at 20.0 MPa and 450 °C. Calculate the percent yield if 750 L of
ammonia gas is collected when 21.8 kg of hydrogen gas is reacted with excess
nitrogen gas.
Hint: Write a balanced chemical equation first.
Do not show your work in the space provided.
Record only your final answer with the correct number of significant digits and
the proper units.
Answer:
Next page
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning