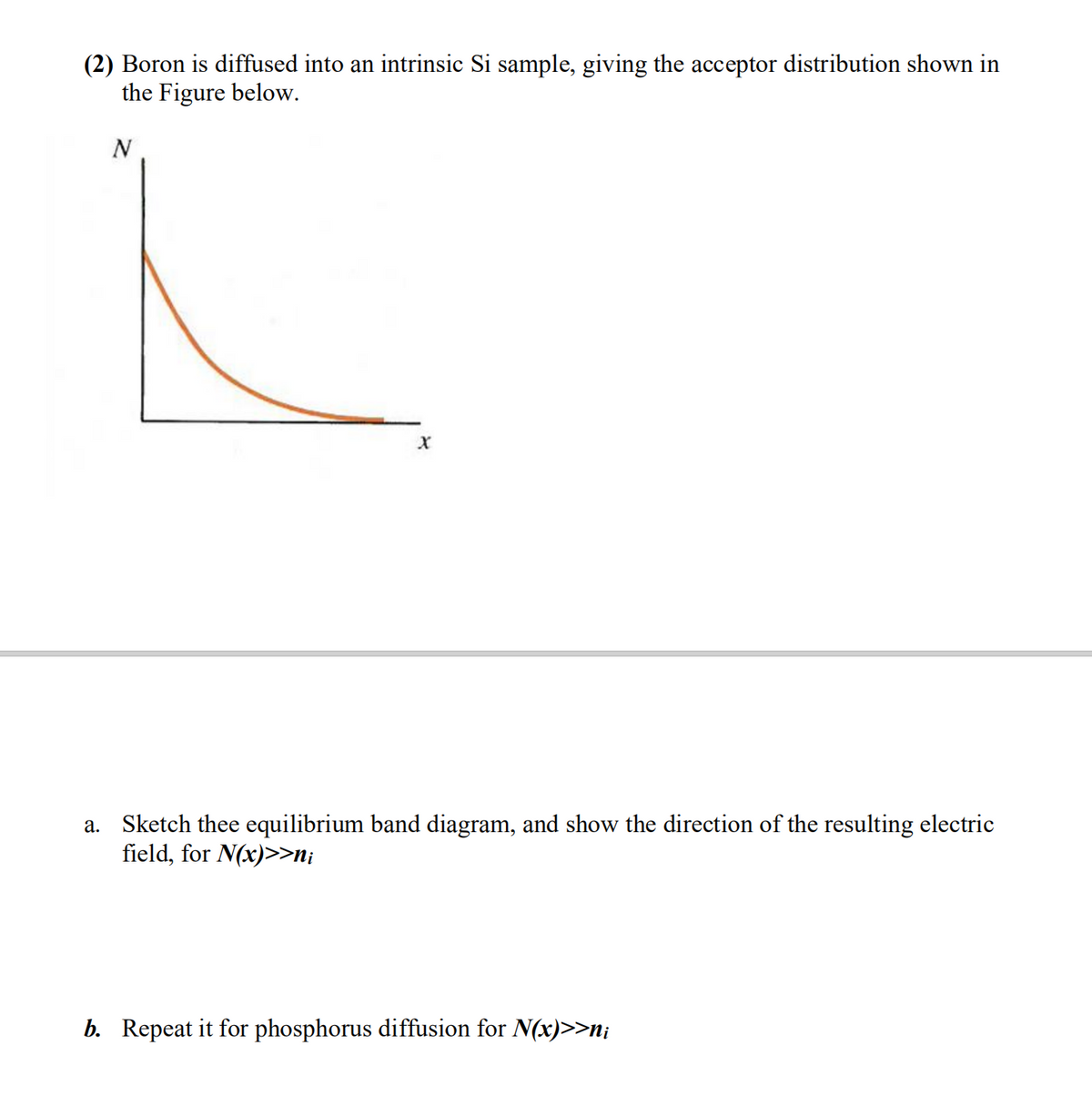
Q: 8. While studying the adsorption isotherm of acetic acid on charcoal, if 2.5 gm of charcoal be taken...
A: Here adsorbate is acetic acid and adsorbent is charcoal. Given the mass of the adsorbent, charcoal ...
Q: Answer the question in full details and complete paragraphs, using lots of information about the top...
A: Polycarbonates are type Of thermoplastics having carbonate groups and are optically Transparent.
Q: 2+ A chemist wishing to do an experiment requiring " Ca* (half-life = 4.5 days) needs 9.0 ug of the ...
A: Given :- Half life of 47Ca2+ = 4.5 da Time = t = 48 hour = 2 day Amount of 47Ca2+ present at time ...
Q: 3. In the conductometric titration, the concentration of alkali is five time stronger than the conce...
A: In conductivity measurements the end point is there where a sharp change of value of conductivity is...
Q: 6. Methanal (HCHO) 7. Naphthalene (CioHe) 8. Acetanilide (CH;-NHCOCH) 9. Uracil NH Cyclohexane (CH12...
A:
Q: 8. Explain why NaCl is soluble in water whereas CaO is insoluble.
A: Strong sodium ions bound to negative chloride ions are made of salt (sodium chloride). Water can dis...
Q: What is the pH of a solution prepared by dissolving 0.450 mol of NH3 in sufficient water to make 1.0...
A: Given, The pH of a solution prepared by dissolving 0.450 mol of NH3 in sufficient water to make 1.0...
Q: The change in the internal energy of a system that absorbs 2,500 J of heat and that does 7,655 J of ...
A: Given-> Heat absorbed = 2500 joule Work done on surrounding = 7655 joule
Q: What is the percent composition of scopolamine, C17H21NO4? __C% __H% __N% __O%
A: Molar mass of C17H21NO4 = molar mass of (17C + 21H + 1N + 4O) = [17(12)+21(1)+1(14)+4(16)] = [204 +...
Q: Which of the following forms of electromagnetic radiation have the longest wavelength? O Radio waves...
A:
Q: The following table shows data from the titration of four 10.0 mL samples of a solution containing h...
A: a.) First we would Calculate volume of NaO* added using formula : Volume of NaOH added = Final bure...
Q: p0 g KHP) and Solution 2 (0.1000 g KHP). What entration of NaOH using Solution 1 comparedt NAOH]: > ...
A: At neutralization point , Equivalents of NaOH = Equivalents of KHP In solution 2 , moles of KHP is h...
Q: consider the reaction: H2(g) + NO(g) arrow forward N2O(g) + H2O(g). What is the total order of react...
A: H2(g) + NO(g) → N2O(g) + H2O(g) the balanced equation is: H2(g) + 2NO(g) → N2O(g) + H2O(g)
Q: 4. The structure of [Cu(NH3)4]²+ is known to be square planar. Using the ligand o orbitals only, wri...
A: Given that Square planer metal complex [Cu(NH3)4]2+ (ML4) Draw orbitals symmetries of the central m...
Q: Calculate the solubility of BaCrO, in water at 25 °C. You'll find K, data in the ALEKS Data tab. sp ...
A: Given-> BaCrO4(Ksp) = 1.17 × 10-10
Q: Write out the net-ionic equation for the precipitation reaction that will happen with hydrogenphosph...
A: When hydrogen phosphate ion , HPO42- and 1 M CaCl2 treated with each other, then following reaction ...
Q: 14) Methanol is the fuel in "canned heat" containers (e.g., Sterno) that are used to heat food at co...
A:
Q: A chemist titrates 180.0 mL of a 0.4289M pyridine (CH,N) solution with 0.5274M HCI solution at 25 °C...
A: Given, [C5H5N] = 0.4289 M Volume of C5H5N in mL = 180.0 mL [HCl] = 0.5274 M pKb of C5H5N = 8.77 ...
Q: If water is added to a 0.70 molar solution of CuSO4, what will change? O Molarity will increase O Mo...
A: Dilution of the solution: When we dilute a concentrated solution with pure water, the mole of solute...
Q: What will be the expected pH of the hydrolyzed aspirin solution after the excess NaOH has been neutr...
A: Aspirin other name is acetylsalicylic acid. Hydrolysis of aspirin means breaking of aspirin by addit...
Q: INSTRUCTIONS: Describe and explain in detail the following Chemical Equilibrium diagram.
A:
Q: Use the References to acc What is the IUPAC name for the following compound? CH3 CH C-CH=CH-CH3
A: Answer of the question given below
Q: 1. Suppose there is an atom of oxygen, containing eight protons and eight neutrons in the nucleus, a...
A: 1. An oxygen atom has eight protons and eight neutrons in the nucleus. Another four neutrons are add...
Q: Calculate the pH of a 0.56 M benzoic acid solution.
A:
Q: average K= 2.3116, Fe = 0.006 please help! 3. Suppose you had prepared Beaker D, where the initial c...
A: Given: Initial concentration of SCN- = 0.0012 M Initial concentration of Fe3+ = 0.006 M And the equi...
Q: Activity 11 This problem is on how to determine the pH of a salt. The salt is NaCIO. Find the pH of ...
A:
Q: For the next 3 items: A 0.498 g sample contains 26.0% aluminum triiodide, Alls, (MW = 407.695) by we...
A: when two soluble salt react it would result in the formation of insoluble salt which does not dissol...
Q: Consider the reaction of 75.0 mL of 0.350 M C;H;N (Kb = 1.7 x 10*) with 100.0 mL of 0.449 M HCI. a) ...
A: Solution Here question has multiple sub parts according to Bartleby guideline we will solv...
Q: Be sure to answer all parts. The equilibrium constant K, for the reaction below is 0.00425 at a cert...
A:
Q: A 22.4 mL sample of 0.155 M HCl is titrated with 0.238 M NaOH. what is the pH after the addition of ...
A:
Q: Consider the I-131 nucleus that is used to treat thyroid. Mass of I-131 is 130.78302 amu. 1) what is...
A: To answer questions regarding I-131 nucleus.
Q: 24) A 1.50 g sample of pure sucrose is burned in a bomb calorimeter that has a heat capacity of 8.93...
A: Mass of Sucrose (m)= 1.50 g Heat Capacity (C) = 8.930 kJ/°C Change in temperature (∆T) = 26.77°C - ...
Q: 8. While studying the adsorption isotherm of acetic acid on charcoal, if 2.5 gm of charcoal be taken...
A: We use the Freundlich equation to plot Freundlich adsorption isotherm curve. Here adsorbate is aceti...
Q: 3. Determine the weight of the sample needed to be mixed to create a 125ml of the folowing compounds...
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for...
Q: What is the percent by mass concentration of NaCl in a solution that is prepared by adding 15.0 g of...
A:
Q: When a different isomer of the starting chloride is used, the exact same product is observed despite...
A:
Q: reaction is studied at various temperatures, and a graph of ln K ( equilibrium constants on y axis) ...
A:
Q: Bb Microso x I Bb Emailin x Bb CHEM 2 X Bb CHEM 2 X с C Expert X b My Que x + Content x Take Te X Wi...
A:
Q: Question 2. Name the compounds A to D (Figure 2.1). Tip: It is necessary to specify the configuratio...
A:
Q: Rubidium-87 decays by B production to strontium-87 with a half life of 4.7 x 1010 years. What is the...
A:
Q: How many moles of acetate ions are pre your answer to four significant figures.
A: 1. number of moles can be calculate by Number of moles (n) = Given mass/ molecular mass 2. Mol...
Q: 2 NOBR (g) = 2 NO (g) + Br2 (g) A) adding NOBr. B) removing NO C) doubling the concentration of NO a...
A: Given- 2NOBr(g) <->2NO + Br2(g)
Q: A chemistry student weighs out 0.186 g of sulfurous acid (H, SO,), a diprotic acid, into a 250. mL v...
A:
Q: Which of the following is/are true about the peptide bond? It has a triple bond character There is ...
A: Amino acids can be defined as the organic compounds containing carboxyl group and amine group.
Q: A chemist titrates 80.0 mL of a 0.1023 M butanoic acid (HC,H,CO,) solution with 0.4765M KOH solution...
A: In this question we will do an acid-base reaction between butanoic acid which is a weak acid and KOH...
Q: The pH of a 0.50 M solution of an acid, HA, is 4.67. Calculate Ka of HA.
A:
Q: 50.00- 45.00 40.00- 36.00 30.00 3 25.00- 20.00- 15.00- 10.00- 5.00 MM CM 0.00 o.bo 1 14.00 200 4.00 ...
A: Peak has the shape of a triangle. Formula for the area of a triangle will be used to calculate the a...
Q: 17. Specific gravity of lipid is А. 0.2 В. О.8 С. 1.0 D. 1.5
A: To identify: Specific gravity of lipid.
Q: According to the video, the unknown compound could be a benzoic acid or mandelic acid. Use the infor...
A: This Technique is Called Mixed melting point Technique in which Unkown is added to Known compounds.
Q: In this experiment, Ag (aq) reacts with SCN (aq) to form a precipitate. Adding AgNO3 (aq), which con...
A: Value of equilibrium constant depends only on the temperature. No other factor can change the value ...
Step by step
Solved in 2 steps with 1 images

- Manganese silicide has the empirical formula MnSi andmelts at 1280 °C. It is insoluble in water but does dissolvein aqueous HF. (a) What type of compound do you expectMnSi to be: metallic, molecular, covalent-network, orionic? (b) Write a likely balanced chemical equation for thereaction of MnSi with concentrated aqueous HF.The fat in a 1.821 g sample of potato chips is extracted with supercritical CO2. After extraction, the residue weighs 1.139 g. What is the fat content (% w/w) of the potato chips?1. A Cr3+ solution is electrolyzed using a current of0.365 A. What mass of Cr(s) (51.9661) is plated outafter 10 hours?2. When an aqueous solution of CuSO4 is electrolyzedcopper metal is deposited. If a constant current waspassed for 5.00 h and 404 mg of Cu (63.546) metal wasdeposited, what was the current?
- what wt. of limestone containing 9.57% Mg must be taken for analysis in order to precipitate of 0.551g Mg2P2O7? how many grams of Na2SO4 are required to ppte Ag2SO4 from 2.000t of AgNO3? a sample of magnetite (impure Fe3O4) weighing 0.5000g is fused with oxidizing flux and the ferric compound formed is eventually precipitated as ferric hydroxide and ignited to ferric oxide which weighs 0.4980calculate %Fe & %Fe2O3A sample of feldspar weighing 1.500g is decomposed, and eventually there is obtained a mixture of KCl and NaCl weighing 0.1801 g. These chlorides are dissolved in H2O, a 50-mL pipetful of 0.08333 N AGNO3 is added, and the precipitate is filtered off. The filtrate requires 16.47 mL of 0.1000N KCNS, with ferric alum as indicator. Calculate the percentage of K2O in the silicate.(c)Ions that react with Ag+ like SCN- can be determined electrogravimetricallyby deposition on a silver working anode:Ag(s) + SCN- ≤ AgSCN(s) + eA silver anode with initial mass of 12.4730 g was used to electrolyse 85.00 mL of asolution of 0.0245 M KSCN. At the end of the experiment, the anode weighed12.5767 g. How long did the experiment take, if a current of 100 mA was used?
- Which of the oxidizing agent listed is (are) capable of oxidizing Br - (aq) to BrO 3 - (aq) in acidicsolutions). Please answer with explanations. Thanks1. What are the practical implications of alloying in carbon and alloy steels? 2. Make use of drawings of segments of the Fe-Cr phase diagram to illustrate the reason for the existence offerritic, martensitic and austenitic stainless steels by also noting the influence of specific alloying elementswhere-applicable. .The chief chemist of the Brite-Metal Electroplating Co. isrequired to certify that the rinse solutions that are discharged from the company’s tin-plating process into the municipal sewer system contain no more than 10 ppm (parts per million) by mass of Sn2+. The chemist devises the following analytical procedure to determine the concentration. At regular intervals, a 100-mL (100-g) sample is withdrawn from the waste stream and acidified to pH = 1.0. A starch solution and 10 mL of 0.10 M potassium iodide are added, and a 25.0-mA current is passed through the solution between platinum electrodes. Iodine appears as a product of electrolysis at the anode when the oxidation of Sn2+ to Sn4+ is practically complete and signals its presence with the deep blue color of a complex formed with starch. What is the maximum duration of electrolysis to the appearance of the blue color that ensures that the concentration of Sn2+does not exceed 10 ppm?



