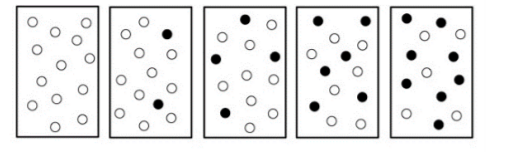
The following diagrams represent a hypothetical reaction A ⇋ B, with A represented by white spheres and B represented by black spheres. The sequence from left to right represents the system as time passes.a. Do the diagrams indicate that the system has reached equilibrium? (Circle one) ( Yes / No )b. Use concentration and reaction rate to describe what is occurring when a system has reached dynamic equilibrium.
The following diagrams represent a hypothetical reaction A ⇋ B, with A represented by white spheres and B represented by black spheres. The sequence from left to right represents the system as time passes.a. Do the diagrams indicate that the system has reached equilibrium? (Circle one) ( Yes / No )b. Use concentration and reaction rate to describe what is occurring when a system has reached dynamic equilibrium.
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter3: Chemical Reactions
Section: Chapter Questions
Problem 13PS
Related questions
Question
The following diagrams represent a hypothetical reaction A ⇋ B, with A represented by white spheres and B represented by black spheres. The sequence from left to right represents the system as time passes.
a. Do the diagrams indicate that the system has reached equilibrium? (Circle one) ( Yes / No )
b. Use concentration and reaction rate to describe what is occurring when a system has reached dynamic equilibrium.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning