The following statements are true for Amperostatic Cuolometric Methods of analysis except - in constant current system, the potential of the cell will vary in time as analyte is consumed cuolometric titrations can be performed at constant current to generate a known quantity of titrant electrochemically a second electrode is no longer needed to detect endpoint of the analysis can be used for acid/base, complexometric and redox titrations The following are true about redox reactions except: * it involves transfer of electrons or charged particles the reaction is performed by bringing the oxidant and the reductant into direct contact in a container the reaction is carried out in an electrochemical cell in which the reactants do not come into contact. an anode is where reduction takes place The following are excitation signals used for voltammetry except-* linear scan differential pulse square wave triangular wavelength quant
The following statements are true for Amperostatic Cuolometric Methods of analysis except - in constant current system, the potential of the cell will vary in time as analyte is consumed cuolometric titrations can be performed at constant current to generate a known quantity of titrant electrochemically a second electrode is no longer needed to detect endpoint of the analysis can be used for acid/base, complexometric and redox titrations The following are true about redox reactions except: * it involves transfer of electrons or charged particles the reaction is performed by bringing the oxidant and the reductant into direct contact in a container the reaction is carried out in an electrochemical cell in which the reactants do not come into contact. an anode is where reduction takes place The following are excitation signals used for voltammetry except-* linear scan differential pulse square wave triangular wavelength quant
Chapter23: Voltammetry
Section: Chapter Questions
Problem 23.11QAP
Related questions
Question
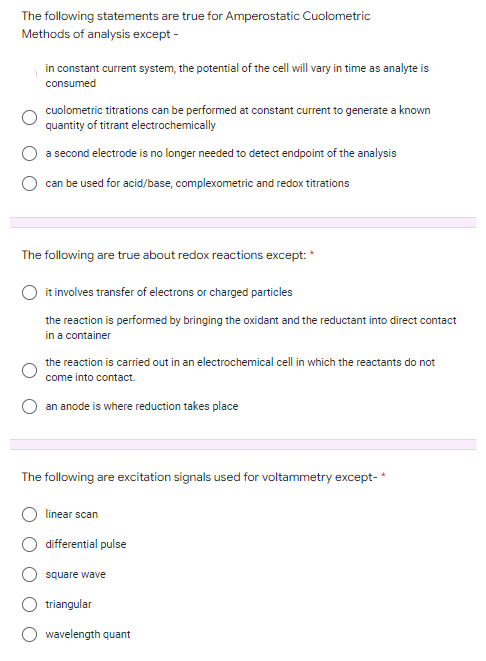
Transcribed Image Text:The following statements are true for Amperostatic Cuolometric
Methods of analysis except -
in constant current system, the potential of the cell will vary in time as analyte is
consumed
cuolometric titrations can be performed at constant current to generate a known
quantity of titrant electrochemically
a second electrode is no longer needed to detect endpoint of the analysis
can be used for acid/base, complexometric and redox titrations
The following are true about redox reactions except: *
it involves transfer of electrons or charged particles
the reaction is performed by bringing the oxidant and the reductant into direct contact
in a container
the reaction is carried out in an electrochemical cell in which the reactants do not
come into contact.
an anode is where reduction takes place
The following are excitation signals used for voltammetry except-*
linear scan
differential pulse
square wave
triangular
wavelength quant
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning