Which of the following statements is false? * O Chromatographic analysis is independent of film thickness Thick films are used with highly volatile analytes because such films retain solutes for a longer time, thus providing a greater time for separation to take place. Thin films are useful for separating species of low volatility in a reasonable length of time. Which of the following is true about a chromatogram? * The signal is plotted as a function of time or volume of the added mobile phase The position of the peak can be used to quantitatively identify the components of the sample The area under the peak provides qualitative measure of the amount of each component The following are characteristics of an ideal GC detector except-* O A temperature range from room temperature to at least 400°C. A long response time independent of flow rate. Adequate sensitivity
Which of the following statements is false? * O Chromatographic analysis is independent of film thickness Thick films are used with highly volatile analytes because such films retain solutes for a longer time, thus providing a greater time for separation to take place. Thin films are useful for separating species of low volatility in a reasonable length of time. Which of the following is true about a chromatogram? * The signal is plotted as a function of time or volume of the added mobile phase The position of the peak can be used to quantitatively identify the components of the sample The area under the peak provides qualitative measure of the amount of each component The following are characteristics of an ideal GC detector except-* O A temperature range from room temperature to at least 400°C. A long response time independent of flow rate. Adequate sensitivity
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter1: Basic Concepts Of Chemistry
Section: Chapter Questions
Problem 62RGQ: You set out to determine the density of lead in the laboratory. Using a top loading balance to...
Related questions
Question
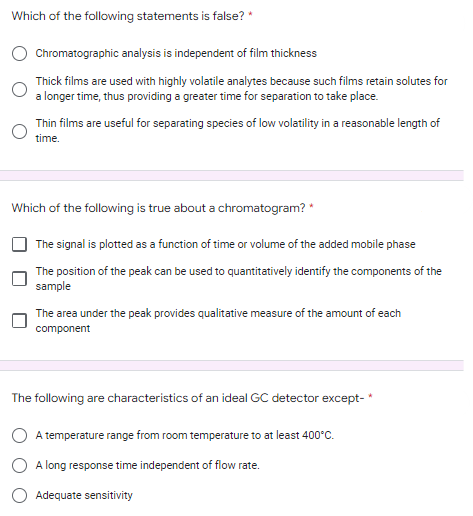
Transcribed Image Text:Which of the following statements is false? *
Chromatographic analysis is independent of film thickness
Thick films are used with highly volatile analytes because such films retain solutes for
a longer time, thus providing a greater time for separation to take place.
Thin films are useful for separating species of low volatility in a reasonable length of
time.
Which of the following is true about a chromatogram? *
The signal is plotted as a function of time or volume of the added mobile phase
The position of the peak can be used to quantitatively identify the components of the
sample
The area under the peak provides qualitative measure of the amount of each
component
The following are characteristics of an ideal GC detector except-*
A temperature range from room temperature to at least 400°C.
A long response time independent of flow rate.
Adequate sensitivity
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning



General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning