The heating element of a coffeemaker operates at 120 V and carries a current of 7.70 A. Assuming the water absorbs all of the energy converted by the resistor, calculate how long it takes to heat 0.252 kg of water from room temperature (23.0°C) to the boiling point. Step 1 The energy required to raise the temperature of an amount of water of mass m, from T, = 23.0°C to the boiling point, T = 100°C, is Q = m C(AT), where the specific heat of water C = 4186 J/kg - °C. We have Q = mFw(AT) = m„„(T – T) 0.252 0.252 kg) (4186 J/kg - °c)(7 77 °C 0.81225 0.812 x 105 J. Step 2 The rate Pat which the heating element converts electrical potential energy into the internal energy of the water is P (AV)I = (120V 120 v7.7 7.7 A- 924 924 J/s. Step 3 Thus, the time At required to bring the water to a boil is |× 105 ) At | J/s) I min 60.0 s min.
The heating element of a coffeemaker operates at 120 V and carries a current of 7.70 A. Assuming the water absorbs all of the energy converted by the resistor, calculate how long it takes to heat 0.252 kg of water from room temperature (23.0°C) to the boiling point. Step 1 The energy required to raise the temperature of an amount of water of mass m, from T, = 23.0°C to the boiling point, T = 100°C, is Q = m C(AT), where the specific heat of water C = 4186 J/kg - °C. We have Q = mFw(AT) = m„„(T – T) 0.252 0.252 kg) (4186 J/kg - °c)(7 77 °C 0.81225 0.812 x 105 J. Step 2 The rate Pat which the heating element converts electrical potential energy into the internal energy of the water is P (AV)I = (120V 120 v7.7 7.7 A- 924 924 J/s. Step 3 Thus, the time At required to bring the water to a boil is |× 105 ) At | J/s) I min 60.0 s min.
Physics for Scientists and Engineers, Technology Update (No access codes included)
9th Edition
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Raymond A. Serway, John W. Jewett
Chapter27: Current And Resistance
Section: Chapter Questions
Problem 27.55P: Review. The healing element of an electric coffee maker operates at 120 V and carries a current of...
Related questions
Question
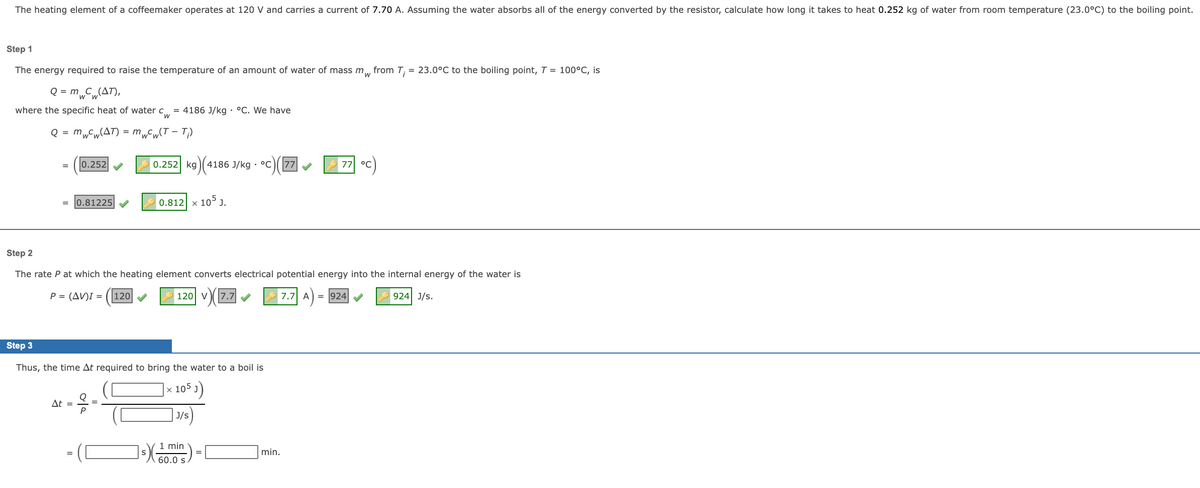
Transcribed Image Text:The heating element of a coffeemaker operates at 120 V and carries a current of 7.70 A. Assuming the water absorbs all of the energy converted by the resistor, calculate how long it takes to heat 0.252 kg of water from room temperature (23.0°C) to the boiling point.
Step 1
The energy required to raise the temperature of an amount of water of mass m, from T;
= 23.0°C to the boiling point, T = 100°C, is
Q = m„C„(AT),
where the specific heat of water c = 4186 J/kg · °C. We have
Q = mwCw(AT) = mwCw(T – T;)
0.252
0.252 kg
4186 J/kg · °C
77
7기 °C
= 0.81225
x 105 J.
0.812
Step 2
The rate P at which the heating element converts electrical potential energy into the internal energy of the water is
P = (AV)I = (|120
120 V
v7.7
7.7 A
= 924
924 J/s.
Step 3
Thus, the time At required to bring the water to a boil is
105 J)
At =
J/s
1 min
min.
60.0 s.
Expert Solution
Step 1
Given information:
The voltage at which the coffee maker works (V) = 120 V
The current it carries (I) = 7.70 A
The mass of the water (m) = 0.252 kg
The initial temperature of the water (Ti) = 230 C
The final temperature of the water required (Tf) = Boiling point = 1000 C
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College


College Physics
Physics
ISBN:
9781305952300
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning