Q: 1.The alcohol content of hard liquor is normally given in terms of the “proof,” which is defined as…
A:
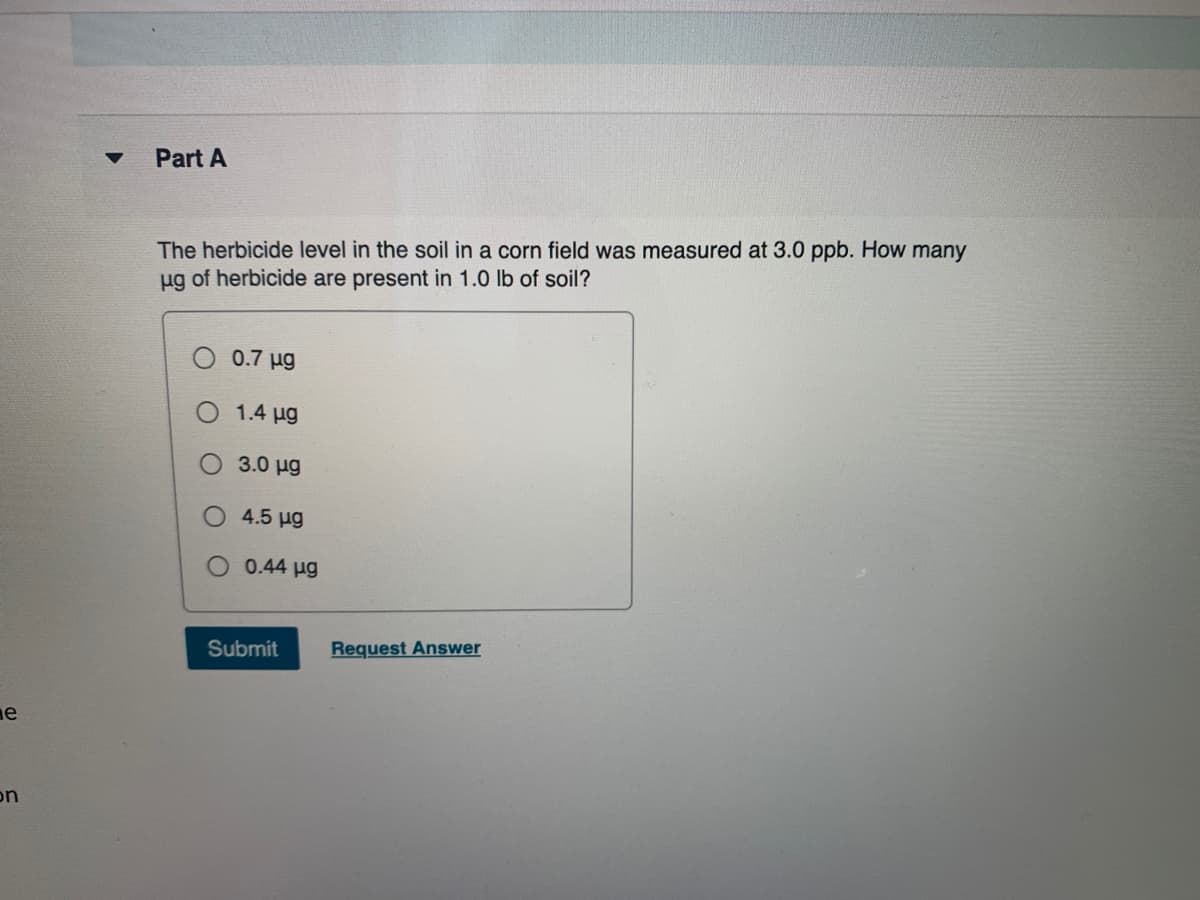
Q: Convert these values 5.0 mg/L seawater to ppm
A: The mass of solute in mg present in a one-liter solution is known as ppm (parts per million)…
Q: 10. A mouthwash contains 22.5% (v/v) alcohol. If the bottle of mouthwash contains 355 mL, what is…
A: Givenv/v percentage of mouthwash= 22.5%Volume of the mouthwash in the bottle= 355mL
Q: THE ACCEPTABLE LEVEL OF COPPER THAT CAN BE FOUND IN DRINKING WATER IS NO MORE THAN 1.3 ppm. A SAMPLE…
A: Given : Safe concentration of copper in drinking water < 1.3 ppm. Mass of water sample = 5000 Kg…
Q: If the specific gravity of methyl salicylate is 1.18, how many grams of methylsalicylate would be…
A:
Q: If a 250 mL glass of water contained 5.00 ppb, what mass of lead is in the glass of water? Assume…
A: The ppb is the form of concentration which can be represented as; ppb=Mass of solute (in g)Mass of…
Q: What is the concentration in ppm of a 3.7 L solution (d = 1.00 g/mL) containing 4.21x10-4g of the…
A: Answer is a) 0.114 ppm
Q: A bubble of air escaping from a diver's mask rises from a depth of 163 ft to the surface where the…
A:
Q: Calculate the ppm of 40.5 mg calcium in 3.50 grams of pill. O 1.16 x10“ ppm 86.4 ppm O 8.64 x107 ppm…
A: Given : Mass of calcium i.e solute = 40.5 mg And mass of pill i.e solvent = 3.50 g
Q: A 50.0-g sample of industrial wastewater was determined to contain 0.48 mg of mercury. Express the…
A: GIVEN: Mass of sample=50.0g Mass of mercury= 0.48 * 10-3g To Solve: mercury concentration of the…
Q: The normal creatinine levels in adults are 0.6-1.2 mg/dL. If a 5.0-mL sample contains 45 μg of…
A: Given that,The normal range of creatinine level in adults is 0.6 to 1.2 mg/dL
Q: A tank contains 6OL of water contaminated with Mercury at a concentration of 0.03 mg/L. Clean water…
A:
Q: 4 mL of a ground water sample from Gallup, NM was found to contain 50 micrograms of U4+ ion. Express…
A: Parts per million (ppm) :- The number of parts by weight of solute present in million (106) parts…
Q: (a) Calculatethe concentration of mercury: (i)in g/L (microgram per liter), and (ii) in mol/L.(b)If…
A: a) Part per billion (ppb) concentration defines the microgram of solute dissolved in 1 L solvent. 1…
Q: Calculate the quantity of each ingredient needed to prepare 15 mL of the following ophthalmic…
A:
Q: How much sugar (C12H22O11) in grams must be added to 450 mL water to make a 20% sugar solution?
A: Density of water = 1 g/mL Mass of water = volume x density = 450 x 1=…
Q: A sample of air analysed at 0°C and 1 atm. pressure is reported to contain 12 ppm of CO. The…
A: ppm can be converted to mg/m3 using the unit conversion method. 1 ppm = 1 mg/L
Q: A water sample contains the pollutant chlorobenzene with a concentration of 15 ppb (by mass). What…
A: Some pollutant chlorobenzene with a concentration of 15 ppb (by mass) is present in water. The…
Q: 64.1 ppm CaCO3 How much calcium would you ingest by drinking one 8 oz glass of your tap water? Show…
A: Given: Concentration of CaCO3 = 64.1 ppm.
Q: Concentration of oxygen dissolved in water* biochemical oxygen demand (BOD) total suspended solids…
A: The amount of oxygen that is dissolved in water is known as dissolved oxygen (DO). The amount of…
Q: 20. Which term best describes the process illustrated here? Semi- permeable membrane Molecule A time…
A: Since the molecules of B is being filtered out from the mixture of A and B in the process. And 1)…
Q: The recommended limit for safe exposure to sulfur dioxide in the air is 0.000025%. What is this…
A: Given, Mass percentage of sulfur dioxide in the air = 0.000025% Then, What is this concentration…
Q: If there is 0.551 mg of arsenic in 348 g of solution, what is the arsenic concentration in ppm?
A:
Q: A drinking water plant adds 500. g of fluoride to a water tank containing 500,000 L of drinking…
A: ppm is the abbreviation of parts per million. It is equivalent to mg/L. Conversion of g into mg: 1…
Q: A bubble of air escaping from a diver's mask rises from a depth of 155 ft to the surface where the…
A: Given the depth of water from the surface, h = 155 ft * 30.48 cm1 ft*1 m100 cm= 47.244 m The volume…
Q: If a single dose of azithromycin for oral suspension for a 1.5 years old child is 125 mg. Calculate…
A: The mass of a single dose of azithromycin for 1.5 years old child is = 125 mg The concentration of…
Q: A 0.1ml serum sample was analyzed for glucose content and found to contain 250 ng. The concentration…
A: Given that: Volume of serum sample = 0.1ml =0.1ml×1L/1000ml = 10-4L Mass of glucose = 250ng =…
Q: Name 10 dissolved substances that negatively affect the environment
A: Dissolved salts may be present in high enough concentrations to be toxic. Eg: naturally occurring…
Q: The concentration of lead in blood samples are often reported in units of micrograms of Pb per…
A: Given : Density of blood = 1 g/mL. And concentration of lead = 60 ppb i.e 60 parts lead per billion…
Q: How many grams of oxygen are required for 100.0 grams of citric acid (C6H8O7) to combust completely…
A: The balanced chemical equation for the combustion of citric acid can be written as follows: C6H8O7…
Q: Glycerin USP( specific gravity 1.25) costs $ 54.25 per pound .if students in a dispensing laboratory…
A: Here the cost of glycerine is $ 54.25 per pound. We have to determine cost of 1 pint glycerine .
Q: A bubble of air escaping from a diver's mask rises from a depth of 159 ft to the surface where the…
A: Density is given as the mass of a substance per unit volume. Density is directly proportional to the…
Q: A pharmacist adds distilled water to 4.00 g of a powdered drug. The final volume of the solution is…
A:
Q: A river pollutant is found to have a concentration of 0.0031 % m/v. If the density of the river…
A: Given %(m/v) = 0.0031 % density of solution = 1.183 gram/mL Concentration of the pollutant in ppm…
Q: What is the volume (in mL) of 2.5 lb benzyl benzoate having a specific gravity of 1.12?
A: Given: Specific gravity = 1.12 mass of benzyl benzoate = 2.5 lb Known, specific gravity =…
Q: he water in a dam has been analyzed to contain 0.35 ppb of Selenium. How many micrograms of Selenium…
A: We have to find out the mass of selenium persent in 10L of water
Q: Grams of plant tissue analyzed and found to contain 92.6 micrograms of zinc. The concentration of…
A: Here we have to find grams of plant tissue (sample) for different PPM given in the question for…
Q: A 2.20−L sample of water contains 165.0 μ g of lead. Calculate the concentration of lead in ppm.
A:
Q: Find the concentration of sodium (in ppm) in a 2.43-g pill that contains 45.0 mg of Na.
A: Given mass of Na = 45.0 mg =0.045 g mass of pill = 2.43 g To find concentration of sodium (in…
Q: Is 10 ppm 400% less than 50 ppm?
A:
Q: How many mL of sterile water are in 3 L of D5W ½ NSS?
A: A numerical problem based on concentration terms, which is to be accomplished.
Q: A bottle of vodka labeled "90 proof" contains 45% (v/v) ethanol in water. How many mL of ethanol are…
A: 45%(v/v) means for 100mL of solution , there is 45 mL of ethanol
Q: How many ppl are there in 1 ppm? 10 100 1000 10000
A: 1000 ppl are there in ppm .
Q: Rubbing alcohol is 35.% (v/v) isopropyl alcohol by volume. How many L of isopropyl alcohol are in a…
A:
Q: A bubble of air escaping from a diver's mask rises from a depth of 152 ft to the surface where the…
A: Given the depth of water, h = 152 ft * 0.3048 m1 ft = 46.33 m density of sea water, d = 1.025 g/mL…
Q: What is the ppm concentration of calcium in a 15 g soil sample if it is found that there is 98…
A:
Q: In Experiment 1, two chemical compounds are introduced. Sodium hydroxide, or NaOH, is a basic…
A: A question based on properties of liquids that is to be accomplished.
Q: A mouthwash contains 22.5% (v/v) alcohol. If the bottle of mouthwash contains 355 mL, what is the…
A: Given,Percent of alcohol in mouth wash = 22.5% (VV).Volume of mouth wash in a bottle = 355mL.
Q: If a room is 6 m long, 5 m wide, and 3 m high. how many milligrams of formaldehyde are present if…
A: Solution stoichiometry involves the calculation of concentration of solutions in the given…
Q: A lake containing 1320g of lead in 31,243,500g of water has what ppm for lead? A. 42.25 ppm B.…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- When drinking water is disinfected with chlorine, an undesired byproduct, chloroform (CHCl3), may form. Suppose a 70-kg person drinks 2 L of water every day for 70 years with a chloroform concentration of 0.08 mg/L (the drinking water standard). Potency factor for chloroform = 6.1 x 10-3 (mg/kg-day)-1 The upper-bound cancer risk for this individual. a. 0.545 b. 5.45x10-6 c. 5.45x10-4 d. 5.45x10-10 e. 14x10-6 If a city with 300,000 people in it also drinks the same amount water described in the above question, how many extra cancers per year would be expected? Assume the standard 70-year lifetime. a. 0.163 b. 1.63x10-4 c. 1.63x10-3 d. 1.63x10-6 e. 6x10-2Oceanic uptake of carbon dioxide is thus described:CO2 (g) + H2O ⇔ H2CO3, K = [H2CO3]/PCO2 = 3 x 10-2 M atm-1 H2CO3 ⇔ HCO3- + H+, K = [HCO3-][H+]/[H2CO3] = 9 x 10-7 moles/LHCO3- ⇔ CO32 - + H+, K = [CO32 -][H+]/[HCO3-] = 7 x 10-10 moles/LCharge balance equation:[H+] = [OH-] + [HCO3-] + 2[CO32 ] If the CO2 concentration in the atmosphere is 300 ppm, what is the pH of the ocean?After Maria’s 8th cup of coffee one night during finals week, she remembered that high doses of caffeine can kill you. Maria was worried that she was more susceptible because she weighed only 47.7 kg. Maria did some research and found that 150 mg/kg of caffeine was lethal and the coffee she was drinking is 0.04% m/v caffeine. How many cups of coffee could Maria drink before she would likely die?
- Oceanic uptake of carbon dioxide is thus described:CO2 (g) + H2O ⇔ H2CO3, K = [H2CO3]/PCO2 = 3 x 10-2 M atm-1 H2CO3 ⇔ HCO3- + H+, K = [HCO3-][H+]/[H2CO3] = 9 x 10-7 moles/LHCO3- ⇔ CO32 - + H+, K = [CO32 -][H+]/[HCO3-] = 7 x 10-10 moles/L CaCO3 ⇔ Ca2+ + CO32- Kc = 4 x 10-9 M2 What is the full charge balance equation for this system?A clinic had 25 patients on Friday morning. If 21 patientswere given flu shots, what percentage of the patientsreceived flu shots? Express your answer to the ones place.The recommended daily dose of calcium for an 18-year-old male is 1200 mg. If 1.0 cup of whole milk contains 290 mg of calcium and milk is his only source of calcium source, how much milk should an 18-year-old male drink each day in mL? Suppose the child is lactose intolerant, do some research and explain what type of milk would be a good substitute for the child. Please give mg of calcium per cup of alternative milk value, and show calculations for how much of the alternative milk an 18-year-old male should drink each day in mL. (Hint: 1 cup = 236.588 mL) Show all work. Give numerical answers in 2 significant figures.
- Table of caffeine standards concentration . Sample Conc, ppm Std1 16 Std2 32 Std3 48 Std4 64 Std5 80 If the volume used to make 100 mL of std 1 is 2 uL what is the concentration in M used to make std calibration curve ? The standards are going to be used to build calibration curve to analyze caffeine in an energy drink. if 500 mL of the energy drink has target of 400 mg , how will you prepare the sample if you need 10 mL for the analysis ? Caffeine MM=194.19 g/mol.Strontium phosphate, Sr3(PO4)2Sr3(PO4)2, dissociates when placed in water. This compound has a solubility of 0.112 mg/L in water at 20 °C. Calculate the Ksp of Sr3(PO4)2Sr3(PO4)2. Please write your answer using scientific notation according to the following format: 0.0010 would be writen as 1.0*10^-3.What is the solubility of HgI2 in water? The Ksp for HgI2 is 4.0X10^-29
- What is the CV of the data below? 2.7 ppm, 3.0 ppm, 2.6 ppm, 2.8 ppm, and 3.2 ppmAbdominal inject a small white rat (18g) with a dosage of 10mg/kg,drug concentration is 0.1%,How much should be injected(ml)?magnesium hydroxide, Mg(OH)2, found in milk of magnesia, has a solubility of 7.05x10-3 gL-1 at 25ºC. Calculate the Ksp for Mg(OH)2 with a tolerance of +8 (Please type answer)



