the isotope needed, by identifying A, . fission reaction: and X, to nce the following lear 235 U+¿n→4Ba+X+3¿n Express your answer as an isotope using prescripts.
the isotope needed, by identifying A, . fission reaction: and X, to nce the following lear 235 U+¿n→4Ba+X+3¿n Express your answer as an isotope using prescripts.
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter18: Nuclear Reactions
Section: Chapter Questions
Problem 13QAP
Related questions
Question

Transcribed Image Text:the isotope needed, by identifying A, .
fission reaction:
and X, to
nce the following
lear
235
U+¿n→4Ba+X+3¿n
Express your answer as an isotope using prescripts.
Expert Solution
Step 1
The process in which nucleus of an atom is divided into two or more smaller, lighter nuclei is known as nuclear fission.
Step 2
The given equation is shown below:
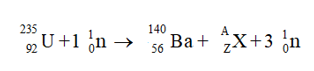
Step 3
The atomic numbers on both sides of the equation must be equal as shown below:
92 + 0 = 56 + Z + 3 (0)
Z = 92 – 56
Z = 36
Step 4
Therefore, the atomic number of X is 36
According to periodic table; the atom which has 36 atomic number is krypton and symbol of that atom is Kr
Step by step
Solved in 7 steps with 3 images

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning
