the link to your ALEKS Hor X McGraw-Hill Education Campus X A ALEKS - Natalie Drury - Learn G Ammonium perchlorate is the so X + lekscgi/x/Isl.exe/1o_u-lgNslkr7j8P3jH-Ugt8PIGunmDn7WsVrRAXK6XnHkiRvH2tl8osQ0ZuornF2Lslfp-YiJWrk55Q4Ewze-XCkvJP7wtwBcw0oPVZ2zD1NgOnWq?1oBw7QYjlbavbSPXtx-YCjsh_7mMmrq#item O CHEMICAL REACTIONS Solving for a reactant using a chemical equation Natalie V Ammonium perchlorate (NH¸C1O,) is the solid rocket fuel used by the U.S. Space Shuttle. It reacts with itself to produce nitrogen gas (N,), chlorine gas (Cl2), oxygen gas (02), water (H,0), and a great deal of energy. What mass of chlorine gas is produced by the reaction of 7.62 g of ammonium perchlorate? Round your answer to 3 significant digits. ola x10 Ar 1II
the link to your ALEKS Hor X McGraw-Hill Education Campus X A ALEKS - Natalie Drury - Learn G Ammonium perchlorate is the so X + lekscgi/x/Isl.exe/1o_u-lgNslkr7j8P3jH-Ugt8PIGunmDn7WsVrRAXK6XnHkiRvH2tl8osQ0ZuornF2Lslfp-YiJWrk55Q4Ewze-XCkvJP7wtwBcw0oPVZ2zD1NgOnWq?1oBw7QYjlbavbSPXtx-YCjsh_7mMmrq#item O CHEMICAL REACTIONS Solving for a reactant using a chemical equation Natalie V Ammonium perchlorate (NH¸C1O,) is the solid rocket fuel used by the U.S. Space Shuttle. It reacts with itself to produce nitrogen gas (N,), chlorine gas (Cl2), oxygen gas (02), water (H,0), and a great deal of energy. What mass of chlorine gas is produced by the reaction of 7.62 g of ammonium perchlorate? Round your answer to 3 significant digits. ola x10 Ar 1II
Chapter16: Applications Of Neutralization Titrations
Section: Chapter Questions
Problem 16.34QAP
Related questions
Question
Transcribed Image Text:the link to your ALEKS Hor X
McGraw-Hill Education Campus X
A ALEKS - Natalie Drury - Learn
G Ammonium perchlorate is the so X +
lekscgi/x/Isl.exe/1o_u-lgNslkr7j8P3jH-Ugt8PIGunmDn7WsVrRAXK6XnHkiRvH2tl8osQ0ZuornF2Lslfp-YiJWrk55Q4Ewze-XCkvJP7wtwBcw0oPVZ2zD1NgOnWq?1oBw7QYjlbavbSPXtx-YCjsh_7mMmrq#item
O CHEMICAL REACTIONS
Solving for a reactant using a chemical equation
Natalie V
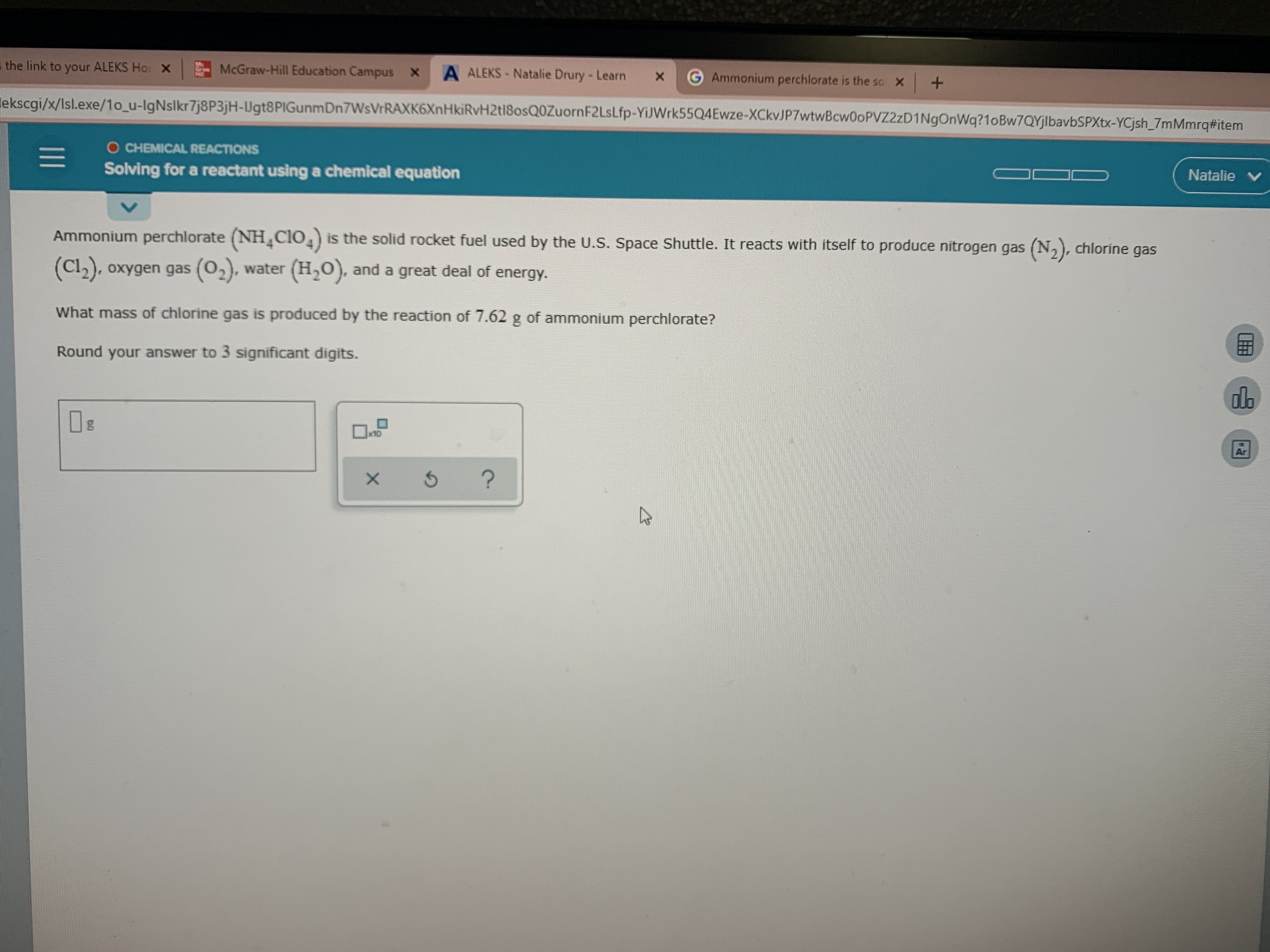
Ammonium perchlorate (NH¸C1O,) is the solid rocket fuel used by the U.S. Space Shuttle. It reacts with itself to produce nitrogen gas (N,), chlorine gas
(Cl2), oxygen gas (02), water (H,0), and a great deal of
energy.
What mass of chlorine gas is produced by the reaction of 7.62 g of ammonium perchlorate?
Round your answer to 3 significant digits.
ola
x10
Ar
1II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning