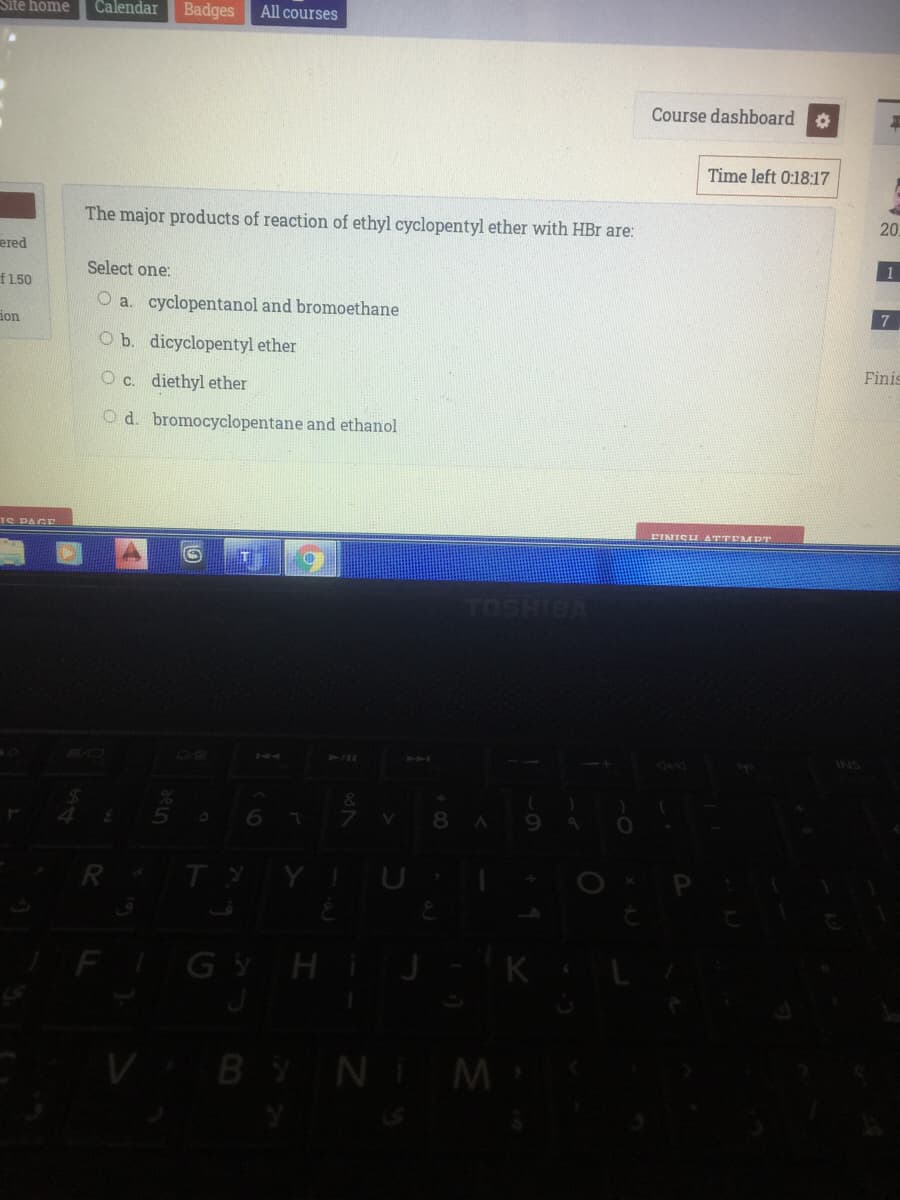
The major products of reaction of ethyl cyclopentyl ether with HBr are: Select one: O a. cyclopentanol and bromoethane O b. dicyclopentyl ether O c. diethyl ether O d. bromocyclopentane and ethanol
The major products of reaction of ethyl cyclopentyl ether with HBr are: Select one: O a. cyclopentanol and bromoethane O b. dicyclopentyl ether O c. diethyl ether O d. bromocyclopentane and ethanol
Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 41MP: In the Gatterman-Kochreaction, a formyl group (—CHO) is introduced directly onto a benzene ring....
Related questions
Question
Transcribed Image Text:Site home
Calendar
Badges
All courses
Course dashboard
Time left 0:18:17
The major products of reaction of ethyl cyclopentyl ether with HBr are:
20
ered
Select one:
f1.50
O a. cyclopentanol and bromoethane
ion
7
O b. dicyclopentyl ether
O c. diethyl ether
Finis
O d. bromocyclopentane and ethanol
TS PAGE
EINISH ATTEMPT
waiHSON
BO
K4
414
INS
&
6.
V.
8
R TYYIU
FI
GYHIJ
VBYN M
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

