Lipids
The heterogeneous classes of organic compounds that are not water-soluble but are dissolved in organic solvents that are non-polar in nature are termed lipids. They are a long chain of fatty acids and esters of alcohols. Lipids are generally seen in several plants, microorganisms, and animals. They are utilized as insulation, components of the cell membrane, hormones, and molecules for the storage of energy.
Glycerophospholipid
Glycerophospholipid is the most abundantly occuring phospholipids found in the biological membranes. Lipids include a group of organic compounds like fats, hormones, oils, waxes, vitamins etc. They are non-polar molecules and are insoluble in water. Lipids play an important role in biological systems. They are the building blocks of our cell membranes, store energy and are involved in signaling.
Structure Of Camphor
A terpene with the molecular formula of C10H16O is a waxy, white color solid known as camphor. It is flammable. It also possesses a very pungent taste and a strong odor. There are various sources for extracting camphor from natural products such as the wood of the tree of camphor laurel. Sublimation of wood and steam distillation are some of the methods involved in obtaining camphor.
Glycolipid In Organic Chemistry
Glycolipids are lipids that are an important class of organic compounds in chemistry that have simple to complex applications. They contain carbohydrates, fatty acids, sphingolipids or a glycerol group. In other words, they are the modifications of lipids like acylglycerols, prenols and ceramides. They are all part of a wider group of compounds known as glycoconjugates.
Diterpenoid
The terpenoid class includes diterpenoids, which are chemical compounds with 20 carbon atoms. They are made up of four isoprene units and are derived from geranylgeraniol, a C20 precursor. They have a C20H32 basic structure. These characteristics distinguish diterpenoids from simple terpenes, which have just 10 carbon atoms.
The carbonyl group was increased acidity compared with
a. false
b. true
Acidic character means compound has ability to donate proton easily.
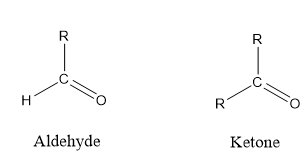
Carbonyl group is classified as group having double bond between carbon and oxygen mainly, aldehyde or ketone.
General formula of aldehyde and ketone groups are RCHO and RCOR respectively.
R is any alkyl group.
Alkene is the molecule having double bond.
Example: Ethene.

Acidic nature of carbonyl and alkene group.
Carbonyl groups are more acidic than alkene because its conjugate base forms resonating structure while in case of alkene not form.
If the stability of conjugate base is higher that means molecule is more acidic.
Higher the resonating structure of conjugate base higher will be the stability of conjugate base.
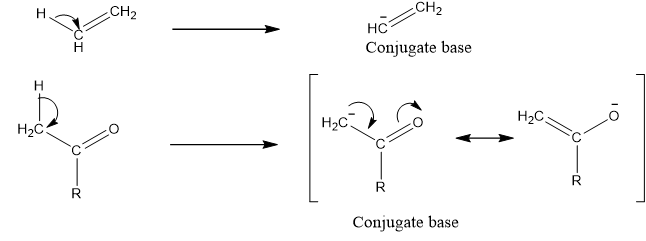
Conjugate base of carbonyl group formed has more resonating structure than ethene therefore, it is more stable and more acidic than ethene.
Step by step
Solved in 3 steps with 3 images




