The mass of solute per 100 mL of solution is abbreviated as (m/v). Mass is not technically the same thing as weight, but the abbreviation (w/v) is also common. How many grams of sucrose are needed to make 625 mL of a 36.0% (w/v) sucrose solution? mass: g sucrose
The mass of solute per 100 mL of solution is abbreviated as (m/v). Mass is not technically the same thing as weight, but the abbreviation (w/v) is also common. How many grams of sucrose are needed to make 625 mL of a 36.0% (w/v) sucrose solution? mass: g sucrose
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter1: Matter And Measurements
Section: Chapter Questions
Problem 53QAP: Magnesium sulfate (MgSO4) has a solubility of 38.9 g/ 100 g H2O at 30C. A solution is prepared by...
Related questions
Question
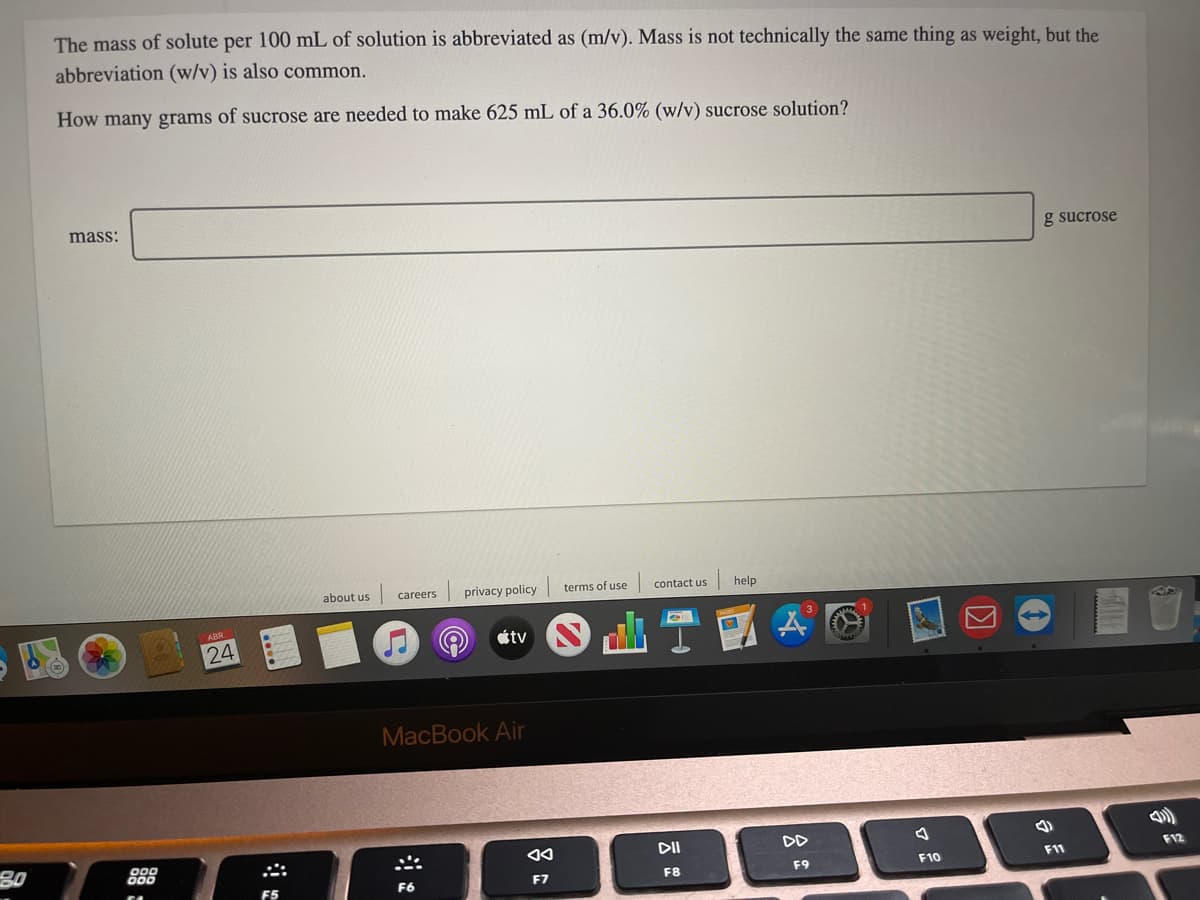
Transcribed Image Text:The mass of solute per 100 mL of solution is abbreviated as (m/v). Mass is not technically the same thing as weight, but the
abbreviation (w/v) is also common.
How many grams of sucrose are needed to make 625 mL of a 36.0% (w/v) sucrose solution?
mass:
g sucrose
about us
careers
privacy policy
terms of use
contact us
help
24
étv
MacBook Air
80
DII
DD
F12
F9
F10
F11
F6
F7
F8
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning