Q: Predict/draw the major product of the reaction shown in the picture:
A: The aromatic compound reacts with bromine from bromo arenes. The reaction proceeds by the aromatic…
Q: Distinguish following compounds by chemical methods: 0 H H o :0
A:
Q: Iron metal reacts with Oxygen gas to produce Iron (III) oxide according to the reaction. AH= -1652…
A: Here we are requires to find the energy released when iron is burnt in oxygen to form iron (III)…
Q: Each row of the table below describes an aqueous solution at 25 °C. The second column of the table…
A: Given, The effect of change on pH when add acid, base and neutral compound to initial solutions is:
Q: & protein in a solution measured in a 96 well plate You have the following information: volume in…
A:
Q: The base protonation constant K of azetidine (C²H²NH) is 1.5 × 10¯8. Calculate the pH of a 0.39 M…
A: The base protonation constant Kb of azetidine C3H6NH is 1.5 × 10-8 .We have to calculate the pH of…
Q: alculate the emf of the cell: Cd | CdCl(aq) I HNO,(aq)| H₂(g) | Pt ) 0.40 V b) -0.40 V c) 0.80 V 311…
A:
Q: Which one of the following substances should exhibit hydrogen bonding in the liquid state? O PH3 O…
A: Molecule is made up of two or more than two atoms.
Q: The mass spectrum of a compound allows us to determine O the molecular weight of the compound O the…
A: A multiple choice question based on mass spectrum that is to be accomplished.
Q: Question 16 of 20 Predict the products (if any) that will be formed by the reaction below. If no…
A: Higher reactive metal can reduce the metal ion of lower reactive metal from its aqueous solution.
Q: For a particular redox reaction, Cr is oxidized to CrO2- and Fe³+ is reduced to Fe²+. Complete and…
A:
Q: Iron metal reacts with Oxygen gas to produce Iron (III) oxide according to the reaction. AH= -1652…
A: Given that , DelH = -1652 KJ The balanced chemical reaction is given by , 2 Fe(s) + 3 O2(g)…
Q: Which of the following complex ions is Paramagnetic (has unpaired electrons)? Explain. a) weak…
A: Paradigmatic substances are those which has unpaired electrons and geomagnetic substances are those…
Q: same compounds. and (a) 0-H O +0-H (c) and 0- CH₂-C-H (e) CH₂=C-H and CH₂-C-H
A:
Q: Which of the following statements are true regarding formaldehyde manufacture: (Multiple answers…
A:
Q: The electrolysis of aqueous Rb2SO4, solution using carbon electrodes produces changes in the…
A:
Q: 1) Supply the structure, name and three letter abbreviation for amino acids which match the…
A: Solutions Amino acids containing side-chains.
Q: CH3- You Br Provide a name for each of the following CH,CH,CH
A:
Q: A toxicologist study about mustard gas, S(CH₂CH₂Cl)2, which is known as a blistering agent. He…
A:
Q: 2. Cl₂ + 2NaBr 2NaCl + Br₂ Oxidation Reaction: Reduction Reaction: Reducing Agent: Oxidizing Agent:
A: Introduction : Redox is a reaction in which a matter gets oxidised mean while the other matter is…
Q: I need help with problem #4
A: Given Synthesis the following reactions Answer only question no 4 as you ask According to the…
Q: pH has an effect on the this/these mode/s of chromatography
A: Ans: HILIC
Q: polar part(s) of the molecule by indicating δ+ and δ- in the appropriate locations and indicate the…
A: REQUIREMENT FROM QUESTION: The polar parts of the molecule. Dipole movement. δ+ and δ- in the…
Q: 15. At 25°C the limiting ionic conductivity of F" is 5.54 mS m² mol', what is its mobility (m V-¹…
A: Formula used : Ionic mobility = Ionic conductance ÷ 96500
Q: For which of the following species are the dispersion forces strongest? C8H18 O C6H14 O C4H10 O C3H8
A: Here C8H18 have highest or strong dispersion forces . Dispersion forces simply vandeerwal…
Q: Br-Cl hv CI Br
A: Requirement from question: Mechanism of given reaction. Initiation, propagation, termination steps.
Q: Which is the best method for the following transformation? OH A. 1. BH3, THF 2. H₂O₂, OH- B. H¹, H₂O…
A:
Q: Which statement is false? O A gas is composed of molecules that are separated from each other by…
A: Postulates of kinetic theory of gases, • Gases are composed of molecules separated from each other…
Q: Question 1 Are the compounds shown below enantiomers, diastereomers, or the same compound? HO....…
A: The molecules with the same molecular formula but different structures are known as isomers. If the…
Q: Write the empirical formula corresponding to C₆H₄Cl₂
A: Empirical formula of a compound is the smallest whole number ratio of elements in the compound.
Q: Directions: Give the oxidation numbers of the underlined atoms in the following molecules and ions.…
A: Sum of oxidation number of each atom is equal to zero. In case of charge, it is equal to charge. Let…
Q: Which reaction will not give the indicated product? 1. CH, CHO 2. H₂O OH O CH₂CH₂OH, H* N H OH…
A: Check the options by the mechanism process.
Q: Give condensed and developed formula to the following polymers 2-methyl, 3-carboxyl pentene 3,3…
A: Let us discuss the structure of each given compounds.
Q: What is the relationship between the following two compounds? SH ОН OH O not isomers; different…
A: Both are same compound.
Q: Screen Shot 2022-06-15 at 19.35.... e O You would like to calculate the concentration of a protein…
A:
Q: ||| H H CH3 Which of the following wedge-and-dash structures represents the chair structure shown…
A:
Q: What is ∆Gο at 25° for the reaction: 2 CO (g) + 2 H2 (g) → CO2(g) + CH4(g), given that at 25° ∆Hο =…
A: Recall data, ∆H°=-247.3 KJ mol-1 ∆S°=-256.5 J mol-1K-1= -0.2565 KJ mol-1K-1 ∆G°=?
Q: The validation process comprises four main steps that are : hardware validation, software…
A: Validation in analytical chemistry is the process of verifying that a procedure yield acceptable…
Q: ncreasing the concentration of the salt in a colloidal system will: a) Increase the radius of sheer.…
A: In question give which option increasing the concentration of the salt in a colloidal system will.…
Q: 1. Determine the oxidation number of Phosphorus in the following Show full calculations. a. Na PO.…
A:
Q: Mode of separation where the mobile phase can be composed of aqueous and organic phases. IEX NPLC…
A:
Q: *There is an ideal solution at 293k. The vapor pressure of material A is P = 500.0 mmHg. And the…
A:
Q: Provide the correct IUPAC name for the compound shown here. Br Br 1 1 CH3-CH-CH-CH3
A:
Q: CH3CH₂COCI AICI3
A: When phenoxy benzene is treated with propanoyl chloride in presence of aluminium chloride, then…
Q: Which of the following complex ions has geometric isomers? explain a) [Co(NH3)6]³+ b) [Co(en)3]³+ c)…
A: For geometrical isomerism all the ligand should not be same.
Q: 2H₂S(g) 2H₂(g) + S₂(g) Kc = 0.0160 What is the value of Kc for the reaction below? 4H₂(g) + 2S₂(g) =…
A:
Q: The rate of radioactive decay follows first order kinetics. Uranium-238 decays very slowly with a…
A: Given, Half-life period (t1/2) = 4.47 billion years Time period = t = 13.4 billion years What is…
Q: rate=] e=k [N₂] ² [H₂]² Use this information to answer the questions below. What is the reaction…
A: We had given rate law, Rate, r = k[N2]2[H2]2
Q: Which chemical equation below is balanced? EQUATION A: 3S8(s) + 16Al(s) ----> 8Al2S3(s)…
A:
Q: Consider the diagram of galvanic cell, which consists of Ag*(aq)/Ag(s) and Sn²+ (aq)/Sn(s)…
A:
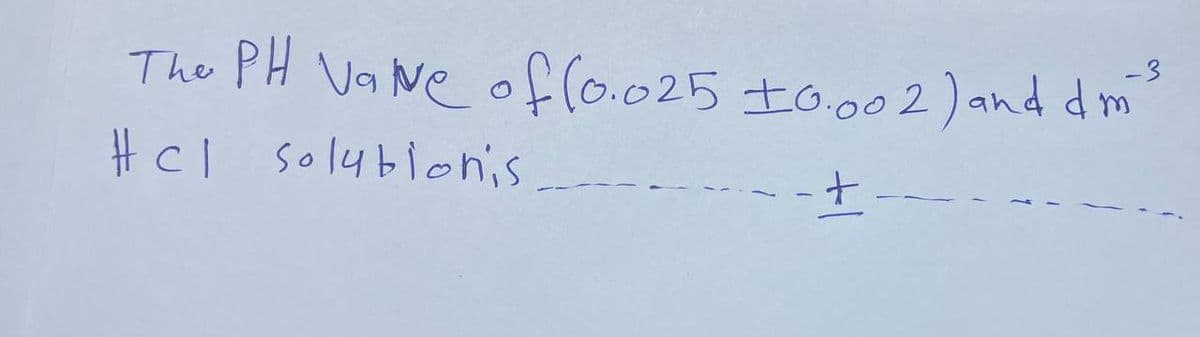
Step by step
Solved in 2 steps with 2 images

- A pH probe/meter uses the following equations: Ecell = L + 0.0592 log a1 = L - 0.0592 pH Where L = L1 + EAg/AgCI + Easy= constants L1 = - 0.0592 log a2 a1 = activity of analyte solution a2 = activity of internal solution Questions: How will measured pH value be affected vs “real” pH if the temperature of the sample is 30C when pH was measured? How will measured pH value be affected vs “real” pH if HCl in pH electrode, became 0.15M instead of 0.1M? How pH value will be affected vs “real” pH if the glass of the pH electrode is not fully hydrated? Please answer all questions and provide a brief explanationCalculate the PH of the solution to 2 significant figures 0.021 moldm-3 HCl show all working outsWhat are the steps to yield Only typed solution
- Chemistry Potentiometry Topic Briefly discuss the following: 1.different types of membrane indicator electrode 2.how a pH glass electrode works 3.errors encountered in the use of pH electrode 4.contribution of other variables in the glass indicator potentialFor an insoluble metallic salt, Ksp will always be less than 1. True or False.Please determine how to make a working standard Solution of about 0.010mg/mL. Caffeine will be the active. Diluent will be water.
- List the advantages and disadvantages of a potentiometric titration relative to a direct potentiometricmeasurement.1. 1093-g sample of impure Na2CO3 was analyzed by residual precipitimetry. After adding 50.00 mL of 0.06911 M AgNO3, the sample was back-titrated with 0.05781 M KSCN, requiring 27.36 mL to reach the endpoint. The percentage Na2CO3 (MW = 106.0 g/mole) in the tested sample is ________ % ? Note: Express final answer using least number of significant figures. 2. The alkalinity of natural waters is usually controlled by OH- (MW = 17.01 g/mole), CO3-2 (MW = 60.01 g/mole), and HCO3- (MW = 61.01 g/mole), which may be present singularly or in combination. Titrating a 10.0-mL sample to a phenolphthalein endpoint requires 38.12 mL of a 0.5812 M solution of HCl, and an additional 18.67 mL of the same titrant to reach the methyl orange endpoint. The composition of the sample is _________% CO3-2 and ___________ % OH- Note: Express final answers using least number of significant figures.Discuss the role of indicators for the titration procedure *
- What is/are the principle/s involved on ACIDIMETRIC ASSAY BY DOUBLE INDICATOR TITRATION?Which has an acitivity coeeficient closest to 1 with ionic strength = 0.001 M? a. Lithium ion b. Magnesium ion c. Lead ion d. Aluminum ionFrom the titration of 50.00 mL of FeSO4 0.2500 mol L-1 with a solution of K2Cr2O7 0.1000 mol L-1 under acidic conditions, in which [H+] = 1.00 mol L-1, calculate the Potential (E) of the system for adding the following volumes of K2Cr2O7 solution: i) 5.00 mL ii) volume at the stoichiometric point (PE) iii) 22.00 mL



