The process in which methylisonitrile is converted to acetonitrile was found to follow first order kinetics. What do the y-intercepts in the graphs below represent for this reaction? 160 5.2 140 120 100 80 60 40 20 0 0 0 10,000 20,000 30,000 Time (s) 10,000 20,000 Time (s) The natural logarithm of the initial pressure of CH3NC The initial rate of reaction The initial concentration of CH3NC The initial concentration of products Pressure, CH₂NC (torr) In pressure, CH₂NC 5445 80 80 1544- 5.0 4.8 4.6 4.4 4.2 4.0 3.8 3.6 3.4 so so c i do 30,000
The process in which methylisonitrile is converted to acetonitrile was found to follow first order kinetics. What do the y-intercepts in the graphs below represent for this reaction? 160 5.2 140 120 100 80 60 40 20 0 0 0 10,000 20,000 30,000 Time (s) 10,000 20,000 Time (s) The natural logarithm of the initial pressure of CH3NC The initial rate of reaction The initial concentration of CH3NC The initial concentration of products Pressure, CH₂NC (torr) In pressure, CH₂NC 5445 80 80 1544- 5.0 4.8 4.6 4.4 4.2 4.0 3.8 3.6 3.4 so so c i do 30,000
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter12: Kinetics
Section: Chapter Questions
Problem 50E: For the past 10 years, the unsaturated hydrocarbon 1, 3-butadiene (CH2 = CH - CH = CH2) has ranked...
Related questions
Question
100%
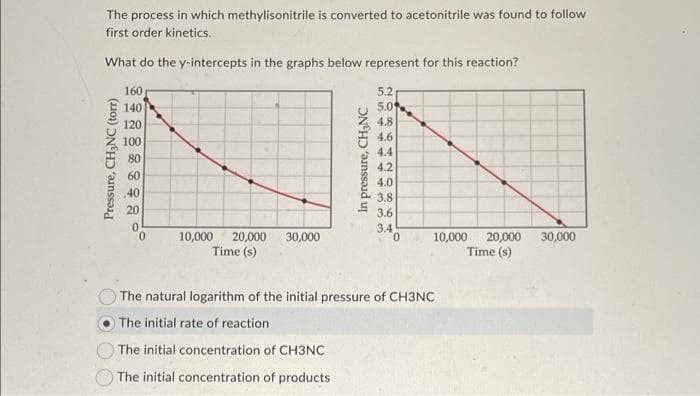
Transcribed Image Text:The process in which methylisonitrile is converted to acetonitrile was found to follow
first order kinetics.
What do the y-intercepts in the graphs below represent for this reaction?
160
5.2
140
120
100
80
60
40
20
0
0
0
10,000 20,000 30,000
Time (s)
10,000 20,000
Time (s)
The natural logarithm of the initial pressure of CH3NC
The initial rate of reaction
The initial concentration of CH3NC
The initial concentration of products
Pressure, CH₂NC (torr)
1544-
29864S
5.0
In pressure, CH₂NC
@@
i do
4.8
4.6
4.4
4.2
4.0
3.8
3.6
3.4
30,000
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning