The rate constant k for a certain reaction is measured at two different temperatures: temperature 376.0 °C 247.0 °C k 5.3 x 10° 2.8× 108 Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy E for this reaction. Round your answer to 2 significant digits. E = 8 kJ mol x10 X
The rate constant k for a certain reaction is measured at two different temperatures: temperature 376.0 °C 247.0 °C k 5.3 x 10° 2.8× 108 Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy E for this reaction. Round your answer to 2 significant digits. E = 8 kJ mol x10 X
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter13: Electrochemistry
Section: Chapter Questions
Problem 13.90PAE: A chemical engineering student is studying the effect of pH on the corrosion of iron. Ellie...
Related questions
Question
G.237.
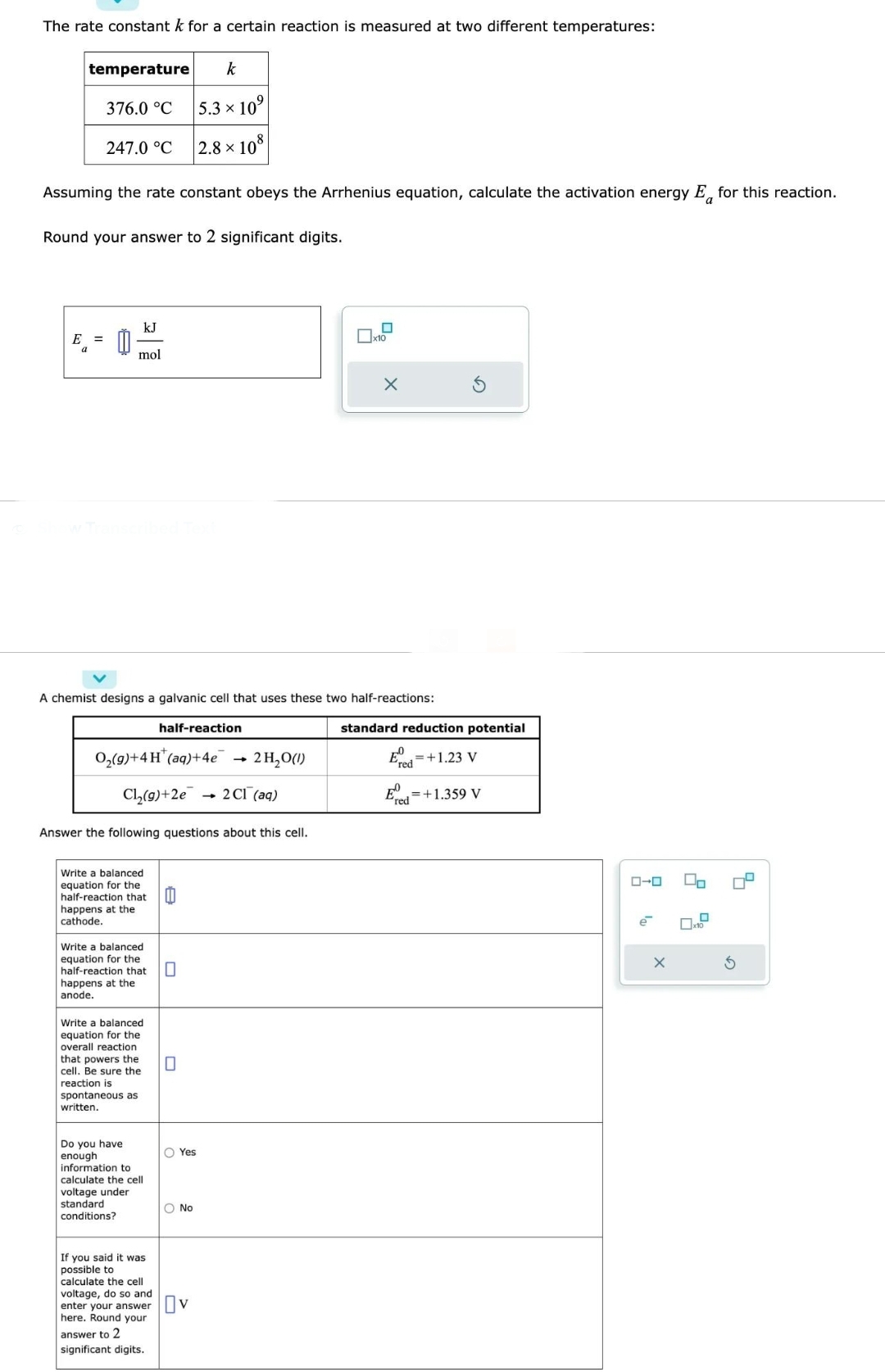
Transcribed Image Text:The rate constant k for a certain reaction is measured at two different temperatures:
temperature
376.0 °C
E =
247.0 °C
Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy E for this reaction.
Round your answer to 2 significant digits.
kJ
mol
A chemist designs a galvanic cell that uses these two half-reactions:
O₂(g)+4H* (aq) +4e
2 H₂O(1)
Cl₂(g)+2e → 2 C1 (aq)
Write a balanced
equation for the
half-reaction that
happens at the
cathode.
Answer the following questions about this cell.
Write a balanced
equation for the
half-reaction that
happens at the
anode.
Write a balanced
equation for the
overall reaction
that powers the
cell. Be sure the
reaction is
spontaneous as
written.
k
5.3 × 10
2.8×108
half-reaction
Do you have
enough
information to
calculate the cell
voltage under
standard
conditions?
If you said it was
possible to
calculate the cell
voltage, do so and
enter your answer
here. Round your
answer to 2
significant digits.
0
O Yes
O No
Ov
x10
X
standard reduction potential
E+1.23 V
red
FO =+1.359 V
red
ローロ
e
00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 7 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning