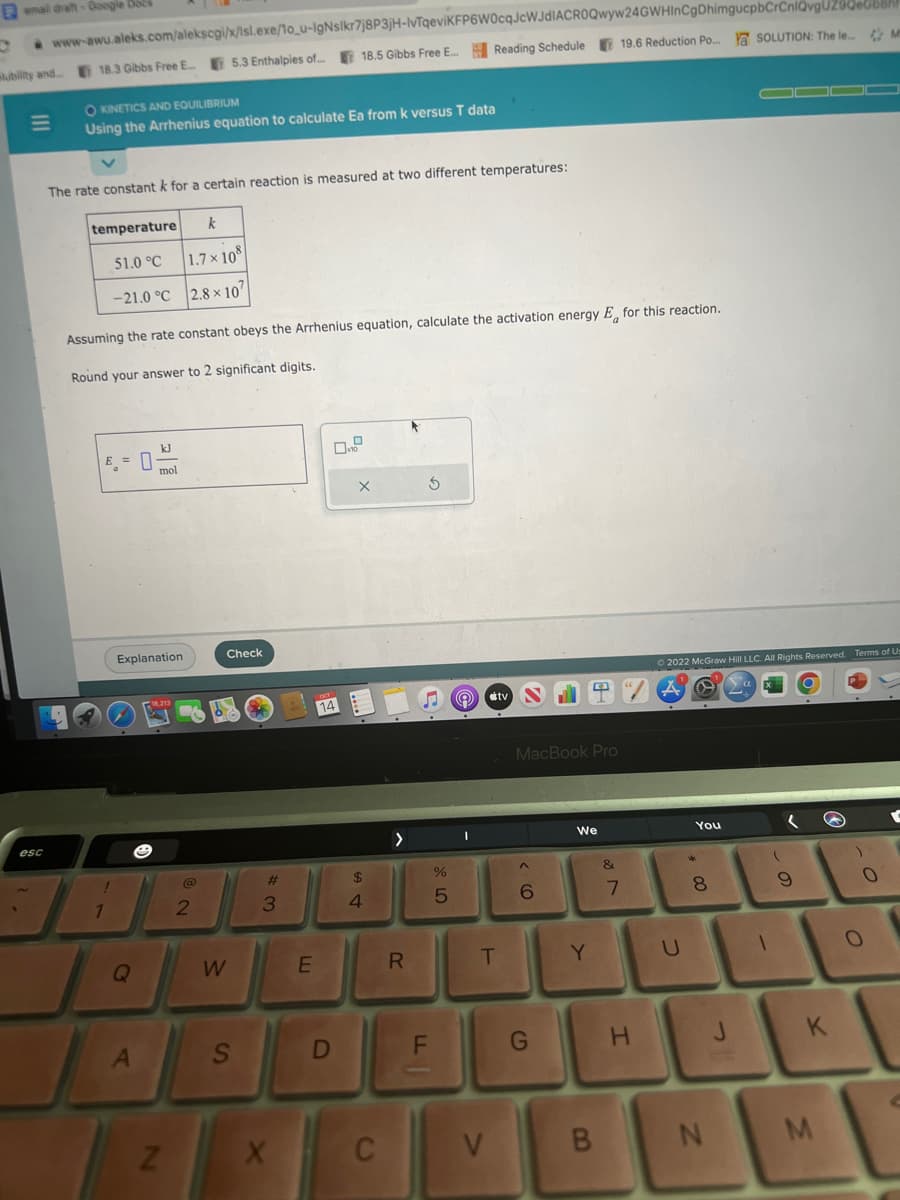
The rate constant k for a certain reaction is measured at two different temperatures: temperature 51.0 °C -21.0 °C Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy E for this reaction. Round your answer to 2 significant digits. E = 0 mol k 1.7×108 2.8 x 107 X G
The rate constant k for a certain reaction is measured at two different temperatures: temperature 51.0 °C -21.0 °C Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy E for this reaction. Round your answer to 2 significant digits. E = 0 mol k 1.7×108 2.8 x 107 X G
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.26QAP
Related questions
Question
Transcribed Image Text:emaill draft-Google Docs
D www-awu.aleks.com/alekscgi/x/lsl.exe/10_u-IgNslkr7j8P3jH-IvTqeviKFP6W0cqJcWJdIACROQwyw24GWHInCgDhimgucpbCrCnlQvgUz9deGbbl
slubility and...
18.5 Gibbs Free E... Reading Schedule 19.6 Reduction Po....
III
E
esc
5.3 Enthalpies of...
18.3 Gibbs Free E...
OKINETICS AND EQUILIBRIUM
Using the Arrhenius equation to calculate Ea from k versus T data
The rate constant k for a certain reaction is measured at two different temperatures:
temperature
k
51.0 °C
1.7×108
-21.0 °C 2.8 x 10
Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy E for this reaction.
Round your answer to 2 significant digits.
E =
!
1
Explanation
D
kJ
mol
A
N
2
Check
W
S
#
3
X
LLI
E
14
D
X
$
4
C
>
R
5
F
%
5
© tv
T
MacBook Pro
6
G
4
We
Y
B
&
7
H
Ⓒ2022 McGraw Hill LLC. All Rights Reserved. Terms of Us
You
* 00
8
U
U
SOLUTION: The le....
N
J
1
(
9
6
R
K
M
→
O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

