k (s-1) The rate of a certain reaction was studied at various T(K) emperatures. The table shows temperature (T) and rate 400 0.000108 constant (k) data collected during the experiments. Plot the 420 0.000626 data to answer the questions. 440 0.00310 What is the value of the activation energy, Ea , for his reaction? 460 0.0134 480 0.0511 500 0.175 520 0.547 Ea kJ . mol- 540 1.57 = 560 4.18 580 10.4 What is the value of the pre-exponential factor (sometimes called the frequency factor), A, for this reaction?
k (s-1) The rate of a certain reaction was studied at various T(K) emperatures. The table shows temperature (T) and rate 400 0.000108 constant (k) data collected during the experiments. Plot the 420 0.000626 data to answer the questions. 440 0.00310 What is the value of the activation energy, Ea , for his reaction? 460 0.0134 480 0.0511 500 0.175 520 0.547 Ea kJ . mol- 540 1.57 = 560 4.18 580 10.4 What is the value of the pre-exponential factor (sometimes called the frequency factor), A, for this reaction?
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.104PAE
Related questions
Question
100%
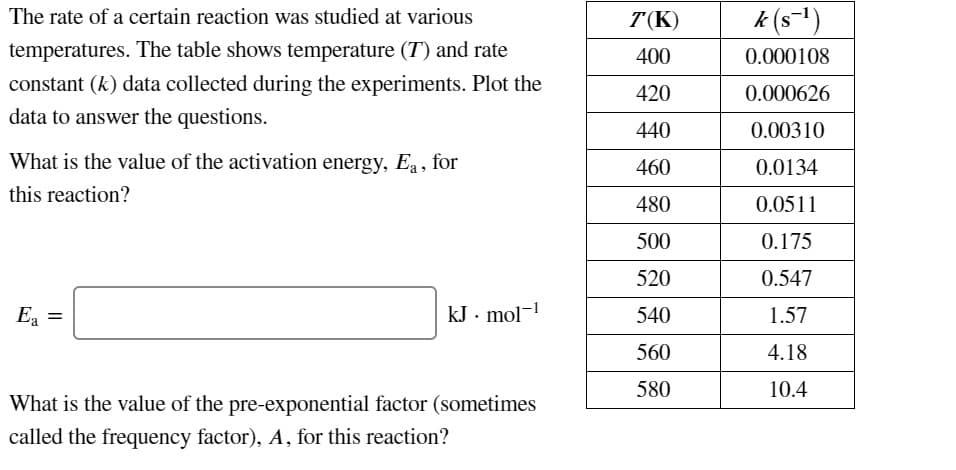
Transcribed Image Text:The rate of a certain reaction was studied at various
T(K)
k (s-1)
temperatures. The table shows temperature (T) and rate
400
0.000108
constant (k) data collected during the experiments. Plot the
420
0.000626
data to answer the questions.
440
0.00310
What is the value of the activation energy, Ea , for
460
0.0134
this reaction?
480
0.0511
500
0.175
520
0.547
Ea =
kJ. mol-
540
1.57
560
4.18
580
10.4
What is the value of the pre-exponential factor (sometimes
called the frequency factor), A, for this reaction?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning