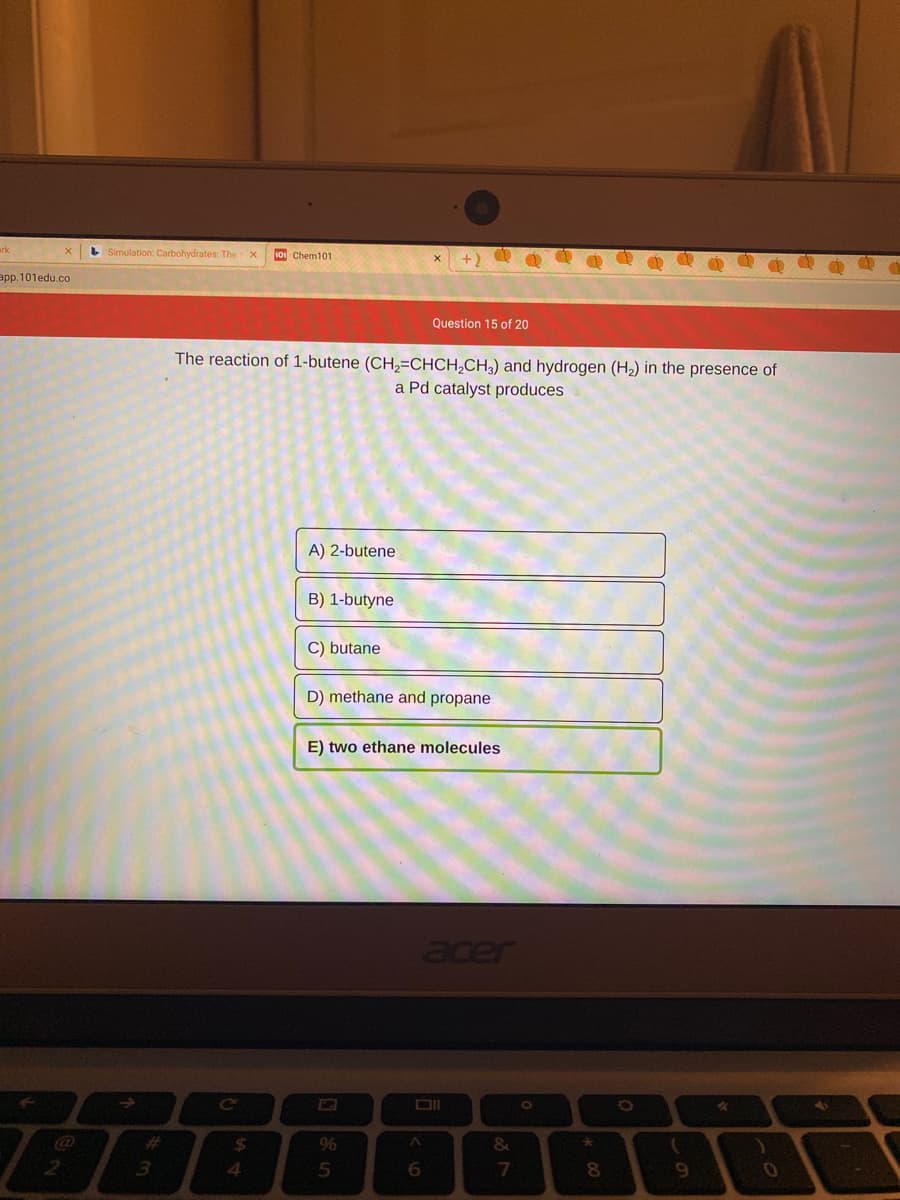
The reaction of 1-butene (CH,=CHCH,CH3) and hydrogen (H2) in the presence of a Pd catalyst produces A) 2-butene B) 1-butyne C) butane D) methane and propane E) two ethane molecules
The reaction of 1-butene (CH,=CHCH,CH3) and hydrogen (H2) in the presence of a Pd catalyst produces A) 2-butene B) 1-butyne C) butane D) methane and propane E) two ethane molecules
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter18: Aromaticity
Section: Chapter Questions
Problem 2CTQ
Related questions
Question
100%
Transcribed Image Text:rk
L Simulation: Carbohydrates: The
10 Chem101
app.101edu.co
Question 15 of 20
The reaction of 1-butene (CH,=CHCH,CH;) and hydrogen (H2) in the presence of
a Pd catalyst produces
A) 2-butene
B) 1-butyne
C) butane
D) methane and propane
E) two ethane molecules
acer
C.
$
%
&
8.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT
