The speed of light is about 3.00 x 108 m/s. How far can light travel in two days (72.0 hours)? Your work/solution for this problem needs to be included in the pages you need to submit for the midterm. Make sure you write the solution on a piece of paper with the appropriate set-up, units and round your answer according to SF. 7.78 x1013 m 5.18 х 1013 m 2.59 x 1013 m 3.00 x 1072 m 5.01 x 1012 m
The speed of light is about 3.00 x 108 m/s. How far can light travel in two days (72.0 hours)? Your work/solution for this problem needs to be included in the pages you need to submit for the midterm. Make sure you write the solution on a piece of paper with the appropriate set-up, units and round your answer according to SF. 7.78 x1013 m 5.18 х 1013 m 2.59 x 1013 m 3.00 x 1072 m 5.01 x 1012 m
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 48E: Although the preferred SI unit of area is the square meter, land is often measured in the metric...
Related questions
Question
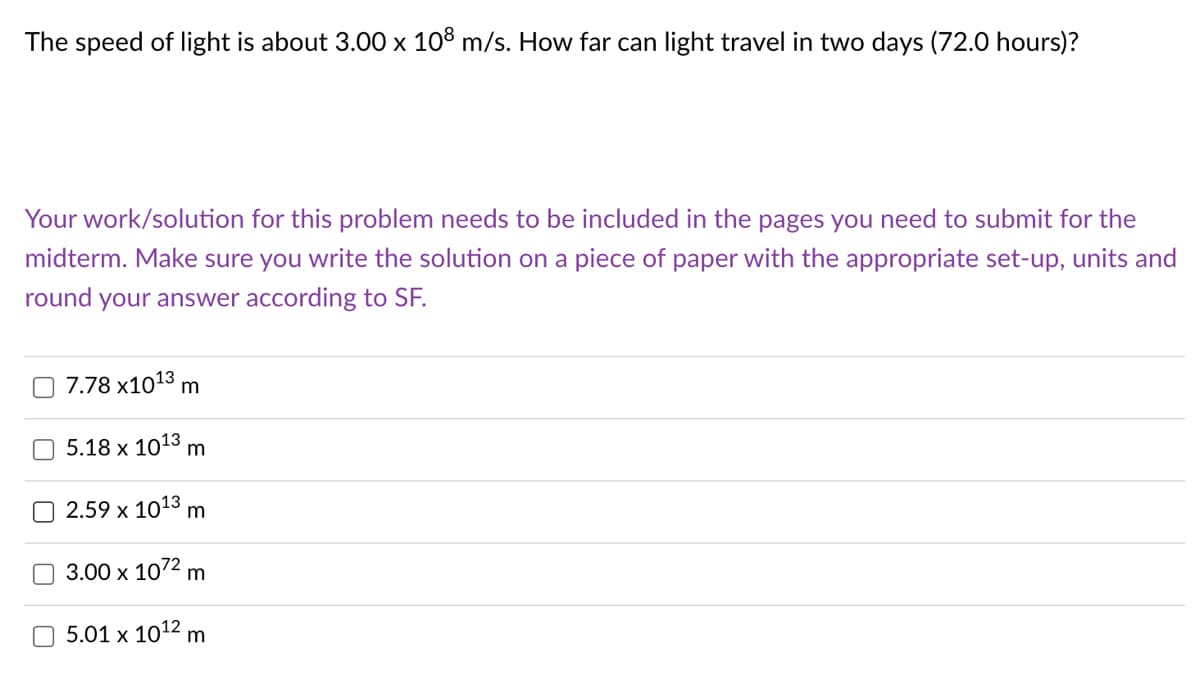
Transcribed Image Text:The speed of light is about 3.00 x 108 m/s. How far can light travel in two days (72.0 hours)?
Your work/solution for this problem needs to be included in the pages you need to submit for the
midterm. Make sure you write the solution on a piece of paper with the appropriate set-up, units and
round your answer according to SF.
7.78 x1013 m
5.18 х 1013 m
2.59 x 1013 m
3.00 x 1072 m
5.01 x 1012 m
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning