Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter19: Eas: Electrophilic Aromatic Substitution
Section: Chapter Questions
Problem 28E
Related questions
Question
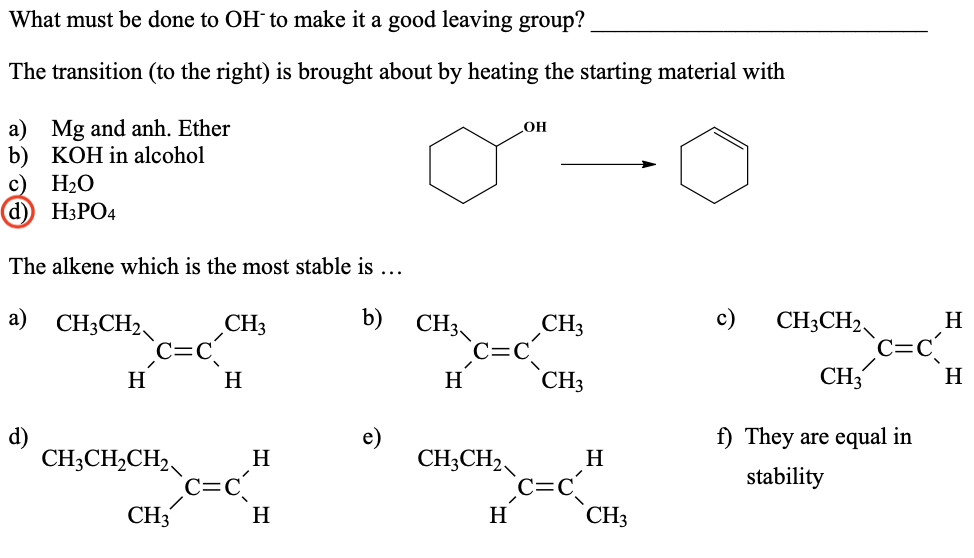
Transcribed Image Text:What must be done to OH´ to make it a good leaving group?
The transition (to the right) is brought about by heating the starting material with
a) Mg and anh. Ether
b) KOH in alcohol
H2O
d) H3PO4
OH
The alkene which is the most stable is ...
a) CH3CH2
b)
с)
CH3CH2
CH3
CH3
CH3
H
H
H
H
CH3
CH3
H
d)
e)
f) They are equal in
CH;CH,CH2
c=c
CH3
CH;CH2
c=c
CH3
H
H
stability
H
H
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
The transition (to the right) is brought about by heating the starting material with a) Mg and anh. Ether b) KOH in alcohol c) H2O d) H3PO4
Solution
Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning