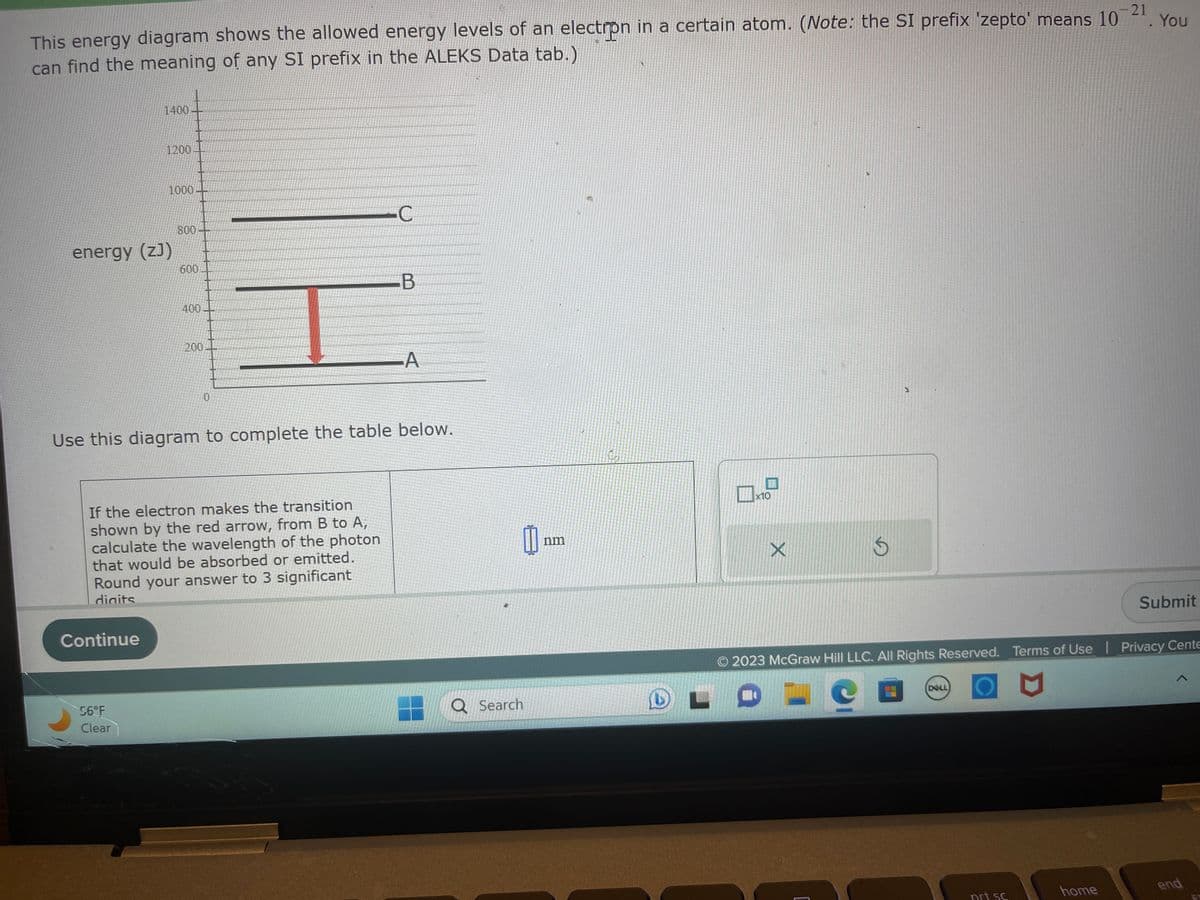
This energy diagram shows the allowed energy levels of an electrpn in a certain atom. (Note: the SI prefix 'zepto' means 10 can find the meaning of any SI prefix in the ALEKS Data tab.) 1400- 1200- 1000. energy (zJ) 800- 600 400- 200 0 C If the electron makes the transition shown by the red arrow, from B to A, calculate the wavelength of the photon that would be absorbed or emitted. Round your answer to 3 significant digits B -A Use this diagram to complete the table below. B nm 10 X S . You Submit
This energy diagram shows the allowed energy levels of an electrpn in a certain atom. (Note: the SI prefix 'zepto' means 10 can find the meaning of any SI prefix in the ALEKS Data tab.) 1400- 1200- 1000. energy (zJ) 800- 600 400- 200 0 C If the electron makes the transition shown by the red arrow, from B to A, calculate the wavelength of the photon that would be absorbed or emitted. Round your answer to 3 significant digits B -A Use this diagram to complete the table below. B nm 10 X S . You Submit
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter7: Atomic Structure And Periodicity
Section: Chapter Questions
Problem 84E: Tbe relative orbital levels for the hydrogen atom can be represented as Draw the relative orbital...
Related questions
Question
Transcribed Image Text:21
This energy diagram shows the allowed energy levels of an electron in a certain atom. (Note: the SI prefix 'zepto' means 10
can find the meaning of any SI prefix in the ALEKS Data tab.)
1400
1200
energy (z))
1000
Continue
56°F
Clear
800
600
400
200
0
If the electron makes the transition
shown by the red arrow, from B to A,
calculate the wavelength of the photon
that would be absorbed or emitted.
Round your answer to 3 significant
digits
Use this diagram to complete the table below.
C
B
A
Q Search
nm
C
x10
X
Ś
DELL
© 2023 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Cente
O
P
prt sc
You
home
Submit
end
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning